Custom Lipoconjugation Services
Molecular imaging has become an indispensable tool in modern diagnostics in biomedical research and therapeutic fields. Thus, the need for highly sensitive and specific
molecular imaging probes is still unmet. In order to design diagnostic imaging and
radiotherapeutic agents, a high target/background ratio, target uptake and rapid
removal of untargeted drug is desirable. Proteins, peptides, antibodies and their
fragments are perfect candidates due to high specificity, affinity and selectivity
to the target organs.
There are various methods commonly used to label proteins, peptides, and antibodies:
- Direct labeling by introducing a radioactive atom to functional groups on the antibody without the use of a chemical spacer. For example, radioiodination through
modification of tyrosine side chains, or
- Indirect method of radiolabeling by introducing chelating agents into biomolecules,which complex with certain radioactive metals.
Types of Bifunctional chelating agents (BCAs) used to create radioimmunoconjugates:
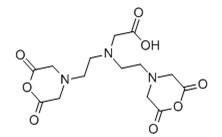
DTPA (Diethylenetriaminepentaacetic Anhydride)
This BCA contains two amine-reactive anhydride groups. DTPA reacts with N-terminal
and ϵ-amine groups of protein to form amide linkage. The anhydride rings open to
create multivalent, metal-chelating arms, capable of tightly binding metals in a
coordination complex. It has 8 coordination sites with metal ions (from 3 nitrogens
and 5 oxygens).
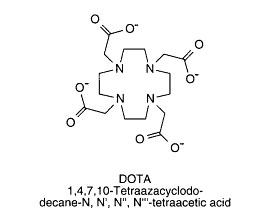
DOTA (1,4,7,10-tetraazacyclododecane-N,N',N'',N'''-tetraacetic acid)
This BCA containing four acetic acid carboxylate groups off the four nitrogens of its twelve atom cyclic structure, is capable of modifying proteins and binding radioactive
metal ions in strong coordination complexes of up to eight dative bonds.
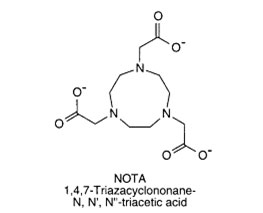
NOTA (1,4,7-triazacyclononane-N,N',N''-triacetic acid)
NOTA has a smaller ring structure in comparison to DOTA and only has three chelating carboxylate groups and three nitrogens, it has six coordination sites with metal
ions.
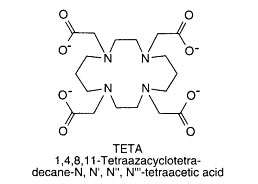
TETA (1,4,8,11-tetraazacyclotetradecane-N,N',N'',N'''-tetraacetic acid)
TETA has four chelating carboxylate groups and four nitrogens. C- and N-functionalized derivatives can be prepare with TETA. A p-bromoacetamidobenzyle-TETA derivative
could be used to label antibodies through sulfhydryl groups.
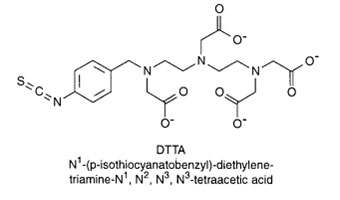
DTTA N-(p-isothiocyanatobenzyl)-diethylenetriamine-N,N',N'',N'''-tetraacetic
acid
This BCA contains four carboxylate groups and three nitrogens that can hold metals
tightly in a coordination complex of seven dative bonds. This compound is good at
chelating lanthanide element series, such as europium, samarium, terbium and dysprosium.This BCA is primarily used for complexing metals to form fluorescent probes for
time-resolved fluoroimmunoassays.
Custom Radioimmunoconjugate Service Descriptions
Sample Submission Requirement:
Biomolecules supplied by customers should be sufficiently pure. Please provide 5
mgs of starting material with the necessary data for purity assessment. Commercially
available biopolymers can be supplied by customers or synthesized and ordered through
Bio-Synthesis.
Price
The price varies depending on the project specifications. The price does not include the cost of small molecule or biopolymer, which is required when ordering
through Bio-Synthesis from a commercial vendor. Some of the small molecules or biomolecules
are commercially available in an activated form. Please contact us for a quote
Macrocyclic Ligands
A series of metal chelating ligands can be covalently attached to biopolymers such as oligonucleotides, antibodies, peptides and proteins. This indirect method of
biomolecule labeling with radiolabels utilizes organic compounds able to chelate
metal ions in a coordination complex. They are widely used in nuclear medicine,
MRI, and optical imaging applications.
Bifunctional Chelating Agent Modification
These bifunctional chelating ligands can be covalently attached to biopolymers such as oligonucleotides, antibodies, peptides and proteins. They are widely used in
nuclear medicine, MRI, and optical imaging applications.
MRI Contrast Agents
Magnetic Resonance Imaging (MRI) contrast agents are a group of contrast media used to improve the visibility of internal body structures using MRI. We offer agents
that are being developed for the ability to measure physiological properties such as pH and temperature.
| Tm-DOTA (Thulium (III) (1,4,7,10-tetraazacyclododecane)-1,4,7,10-tetraacetic acid)
|
| Gd-DOTA Gadolinium (III) (1,4,7,10-tetraazacyclododecane)-1,4,7,10-tetraacetate
|
| Tm[DOTP]5- (Thullium (III) (1,4,7,10-Tetraazacyclododecane-1,4,7,10-tetra(methylene
phosphonate)))
|
| Tm-DOTMA, (Thulium (III) (1R,4R,7R,10R)-α,α',α'',α'''-tetramethyl-1,4,7,10-tetraazacyclododecane-1,4,7,10-tetraacetic
acid
|
| Eu-DOTA-4AmC, Europium (III) [1,4,7,10-tetraazacyclododecane-1,4,7,10-tetrakis(acetamidoacetic
acid)]
|
Chemistry:
Coupling of preactivated small molecules and biomolecules
with chemical reactive groups such as amine, thiol, carboxylate, hydroxyl, aldehyde,ketone, and active hydrogen through the use of various cross linkers.
CAUTION: Conjugation with an antibody may cause some immunoglobulins to lose antigen-binding activity after modification. Pilot experiments to optimize the amount of protein/antibody present and the quantity of metal chelator added to the reaction needs to be done
first in order to avoid this type of crosslinking and polymerization.
Service Specification:
After standard desalting or purification,
a small percent of heterogeneous products containing single or multi-site conjugate per molecule may exist.
Procedure:
After labeling, final conjugates must first be isolated
from excess or unreacted reagent by gel filtration or dialysis. Additional purification techniqies such as stirred cell filtration, tangential flow filtration (TFF), and
gel filtration chromatography may also be used to either remove excess reagent or
isolate and characterize the cross-linked product. For reagents (mostly protein and other biological molecules) that are similar in size or larger than the antibody,one must resort to other purification techniques such as affinity chromatography,ion-exchange chromatography, and hydrophobic interaction chromatography.
Cross-linked target molecules may then be further characterized by biochemical or
biophysical techniques. Once the product has been purified, it may be subject to
many different types of studies including spectroscopic (MALDI-TOF, ESI, LC-MS Fluorescence),
electrophoresis, immunochemical, biochemical, and enzymatical analysis. QC (quality control) and QA (quality assurance) procedures are also followed independently to offer you double guarantee for the highest quality possible. Moreover, our dedicated technical account managers will guide you through every step of the process and constantly keep you informed of the latest project progress.
Ordering and Submitting Requests for Bioconjugation Services
For us to better understand your customized project, please complete our Bioconjugation Service Questionnaire. The more our chemists understand your project’s needs, the more accurate your provided feedback will be. Providing us with your project’s details enables us to recommend the best reagents to use for your project. The most useful and readily available tools for bioconjugation projects are cross-linking reagents. A large number of cross-linkers, also known as bifunctional reagents, have been developed. There are several ways to classify the cross-linkers, such as the type of reactive group, hydrophobicity or hydrophilicity and the length of the spacer between reactive groups. Other factors to consider are whether the two reactive groups are the same or different (i.e. heterobifunctional or homobifunctional reagents), spacer is cleavable and if reagents are membrane permeable or impermeable. The most accessible and abundant reactive groups in proteins are the ϵ-amino groups of lysine. Therefore, a large number of the most common cross-linkers are amino selective reagents, such as imidoesters, sulfo-N-hydroxysuccinimide esters and N-hydroxysuccinimide esters. Due to the high reactivity of the thiol group with N-ethylmaleimide, iodoacetate and a-halocarbonyl compounds, new cross-linkers have been developed containing maleimide and a-carbonyl moieties. Usually, N-alkylmaleimides are more stable than their N-aryl counterparts.
In addition to the reactive groups on the cross-linkers, a wide variety of connectors and spacer arms have also been developed. The nature and length of the spacer arm play an important role in the functionality. Longer spacer arms are generally more effective when coupling large proteins or those with sterically protected reactive side-chains. Other important considerations are the hydrophobicity, hydrophilicity and the conformational flexibility. Long aliphatic chains generally fold on themselves when in an aqueous environment, making the actual distance spanned by such linker arms less than expected. Instead, spacers containing more rigid structures (for example, aromatic groups or cycloalkanes) should be used. These structures, however, tend to be very hydrophobic which could significantly decrease the solubility of the modified molecules or even modify some of their properties. In such cases, it is recommended to choose a spacer that contains an alkyl ether (PEO) chain. Bio-Synthesis offers several cross-linkers with PEO chains, such as thiol-binding homobifunctional reagents, heterobifunctional bases and their derivatives.
Within 3-5 days upon receiving your project scope, we will provide you an appropriate quotation. An order can be placed with PO (Purchase Order) or major credit cards (  ). Your credit card will be billed under Bio-Synthesis, Inc.
). Your credit card will be billed under Bio-Synthesis, Inc.