Disulfide Bridge Cys-Cys Thiol Oxidation
Peptide disulfide bridge readily synthesizes two thiol (SH) groups from the side chain of cysteine or cysteine analogs. Our unique synthesis strategy enables us to offer either specific intra or intermolecular oxidation using appropriate protecting group chemistry to prevent undesired linkage. The reaction can be followed by HPLC and MALDI TOF mass spectrometry with the linear peptide losing two mass units (2H) on cyclization.
In General, disulfide bridge can be formed as follows:
- Intermolecular (two peptide molecules are linked via the disulfide bridge), resulting in either:
- homodimers (two identical peptides) or
- heterodimers (two different peptides).
- Intramolecular (cyclization within one peptide molecule)
While head-to-tail cyclization is usually formed by using an amide bond formation, side-chain-to-side-chain peptide cyclization is most often synthesized via cys-cys or amide bond formation.
Peptide cyclization using cys-cys has limited stability in reductive conditions. Therefore, projects involving drug discovery, commonly use cys-cys cyclized peptides for initial screening and optimization followed by replacement of disulfide bonds with more stable bonds.
Peptides with two or more disulfide bridges require selective protection of the cysteine side chains to ensure that the correct disulfide bridges are formed. We provide peptides with up to 4 disulfide bonds in one peptide using site-specific orthogonal chemistry or thermodynamic stability methods. Contact us to discuss your project details.
Amide Bond formation, Lactam Formation
Cyclic peptides can also be synthesized by linking the amino (N) terminus of the peptide to the carboxyl (C) terminus via an amide bond. The amino side chains of Lys and Orn and the carboxyl side chains of Asp and Glu can also be used to construct cyclic peptides via an amide bond. Amide bonds are more chemically stable than disulfide bridge. Depending
on functional groups of a peptide, cyclic peptide synthesis can be formed in four different ways:
- Head-to-tail between N-terminus and C-terminus
- Head-to-side chain between N-terminus and an internal COOH (e.g. the ß-COOH-group of Asp or γ-COOH-group of
Glu)
- Side chain-to-tail between internal NH2s and C-terminus (e.g. the ε-NH2–group of Lys)
- Side-chain-to-side-chain between an internal NH2 and an internal COOH (e.g. the ε-NH2–group of Lys with either the ß-COOH-group of Asp or γ-COOH-group of Glu)
As with disulfide cys-cys cyclization, amide cyclization is also limited by the challenges of dimerization, undesirable side reactions such as racemization or peptide capping by coupling reagents. For an effective head-to tail cyclization, it is important to carefully select cyclization site, reagent, and optimized cyclization conditions. Bio-Synthesis has extensive experience in synthesizing amide cyclic peptides with a high success rate.
Stapled Peptide Synthesis
Cysteines are common structural motifs in naturally occurring peptides such as neurotoxins, somatostatin and insulin. These disulfide bridges are readily reduced to their acyclic thiol form in an intracellular environment. This challenge can be overcome by using hydrocarbon-stapled peptide synthesis. These peptides are capable of forming stable alpha helical structures as a result of “hydrocarbon stapling" (1,2). Stapled peptides have a chemically locked conformational structure which can mimic the molecular structures that are typically found at the interface of protein-protein interactions. When locked into this stable configuration, constrained peptides are able to penetrate cells and can exert their effect on intracellular protein targets. The large surface area of peptides gives them an advantage over small molecules in their ability to disrupt specific signaling pathways by inhibiting targeted protein-protein interactions. Hydrocarbon stapling is a useful strategy in researching experimental and therapeutic modulation of protein-protein interactions as well as in-vivo pharmacokinetics studies.
References:
- Walensky, LD. et al. Science 305, 1466 (2004)
- Schafmeister, CE. et al. J. Am. Chem. Soc. 122, 5891 (2002)
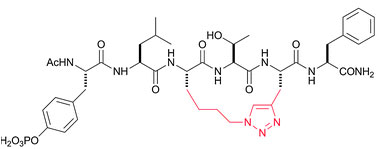
Click Chemistry for peptide cyclization
The cyclization technique increases peptide molecules' potency and in vivo half-life by locking their conformation. In addition to disulfide and amide cyclizations, many other cyclization methods are available at Bio-Synthesis. Clickable functional groups can be incorporated into synthesized peptides using different combinations of protected amino acids modified with an alkyne group, followed by a click reaction with an azido acid. The resulting peptides are detached from the resin to give triazole-containing peptides. Post-synthesis modification allows the introduction of these functional groups to produce structurally constrained peptides. Bio-Synthesis scientists have used click reactions to prepare several different peptide cyclizations.
Please note: Bio-Synthesis does not offer Copper based Click Chemistry, we only offer Copper free click chemistry. For Copper based Click Chemistry please contact baseclick GMBH.
For additional information, please contact us or send an email to info@biosyn.com.