Custom Enzyme-Oligo Conjugation Services
Product Offering:
- Oligo synthesis, functionalization, purification and activation of oligo up to 25 base
- Conjugation of oligo to select enzyme of choice
- Delivery in guaranteed amounts
- Validate by mass spectrometry and SDS gel electrophoresis
There are two ways to place enzyme-oligonucleotide conjugation services. You may request your conjugation services by final set amount of oligo or enzyme starting material.
DNA or RNA can be conjugated to enzyme listed below:
- Horseradish Peroxidase (HP)
- Alkaline Phosphatase (ALP)
- Alkaline Phosphatase Amplification (AMP)
- Penicillinase (PNC)
- DNA-Streptavidin Conjugates
- RNA-Streptavidin Conjugates
- Biotin-Avidin systems
- alpha-D-Galactorsidse
- Glucose-t-Phosphate Dehydrogenase
- Other Enzyme Innunoconjugates
- Nonenzyme protein immunoconjugates
Case Study for Oligo-HRP Conjugate
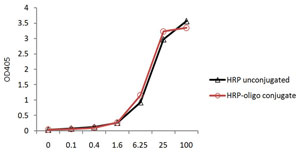
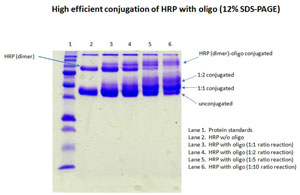
Price
Starting at $800 and up for enzyme-oligo conjugation service. Price varies
from type of enzyme used, amount of starting material or final guaranteed set amount. Please contact us for a quotation.
Service Description
Enzymes maybe modified to contain reactive groups useful for conjugation with oligonucleotides. This can be achieved by using homobifunctional or heterobifunctional cross-linkers that can covalently couple to enzyme and result in terminal reactive group that can cross-link with oligonucleotide containing an amine, thiol and aldehyde modified oligonucleotide.

Specification
Depending on project specification, a pool of heterogeneous products in small percentage may exist.
Procedure
Thiol modified oligo is manufactured under strict, quality control processes. Analytical HPLC and MS analyses are performed in every development cycle. Thiolated oligos are conjugated with SPDP-activated enzyme followed by dialysis, gel filtration or use of the centrifugal concentrator, to remove excess cross-linking reagents and oligonucleotides.
Disclaimer:
Whereas, we can chemically link the intended components of a conjugated molecule, there exists the possibility that the binding sites/active sites of the protein can be altered/modified (partially or completely) independent of the stoichiometry used. Sometimes this activity loss is caused by physically blocking the antigen binding sites, during conjugation or by conformational changes in the complement-determining regions. Some proteins/antibodies are just too labile to undergo chemical modification reactions, regardless of the coupling methods used.
Bio-Synthesis can guarantee the structure of our conjugates but not the suitability to specific biological applications
Ordering and Submitting Requests for Bioconjugation Services
For us to better understand your customized project, please complete our Bioconjugation Service Questionnaire. The more our chemists understand your project’s needs, the more accurate your provided feedback will be. Providing us with your project’s details enables us to recommend the best reagents to use for your project. The most useful and readily available tools for bioconjugation projects are cross-linking reagents. A large number of cross-linkers, also known as bifunctional reagents, have been developed. There are several ways to classify the cross-linkers, such as the type of reactive group, hydrophobicity or hydrophilicity and the length of the spacer between reactive groups. Other factors to consider are whether the two reactive groups are the same or different (i.e. heterobifunctional or homobifunctional reagents), spacer is cleavable and if reagents are membrane permeable or impermeable. The most accessible and abundant reactive groups in proteins are the ϵ-amino groups of lysine. Therefore, a large number of the most common cross-linkers are amino selective reagents, such as imidoesters, sulfo-N-hydroxysuccinimide esters and N-hydroxysuccinimide esters. Due to the high reactivity of the thiol group with N-ethylmaleimide, iodoacetate and a-halocarbonyl compounds, new cross-linkers have been developed containing maleimide and a-carbonyl moieties. Usually, N-alkylmaleimides are more stable than their N-aryl counterparts.
In addition to the reactive groups on the cross-linkers, a wide variety of connectors and spacer arms have also been developed. The nature and length of the spacer arm play an important role in the functionality. Longer spacer arms are generally more effective when coupling large proteins or those with sterically protected reactive side-chains. Other important considerations are the hydrophobicity, hydrophilicity and the conformational flexibility. Long aliphatic chains generally fold on themselves when in an aqueous environment, making the actual distance spanned by such linker arms less than expected. Instead, spacers containing more rigid structures (for example, aromatic groups or cycloalkanes) should be used. These structures, however, tend to be very hydrophobic which could significantly decrease the solubility of the modified molecules or even modify some of their properties. In such cases, it is recommended to choose a spacer that contains an alkyl ether (PEO) chain. Bio-Synthesis offers several cross-linkers with PEO chains, such as thiol-binding homobifunctional reagents, heterobifunctional bases and their derivatives.
Within 3-5 days upon receiving your project scope, we will provide you an appropriate quotation. An order can be placed with PO (Purchase Order) or major credit cards (  ). Your credit card will be billed under Bio-Synthesis, Inc.
). Your credit card will be billed under Bio-Synthesis, Inc.