The last two decades have seen an intensified development of nanoscale calorimetric and electrochemical sensors. Some of these sensors utilize on–target-inducible folding or unfolding of electrode-bound oligonucleotides which may also include aptamers or DNA switches. Examples are sensors for the detection of specific nucleic acids, proteins, small molecules, such as illicit drugs, and inorganic ions. Electrochemical DNA (E-DNA) and electrochemical aptamer-based sensors (E-AB) usually consist of an oligonucleotide probe modified with a redox reporter molecule at one terminal end.
In most cases, the oligonucleotide conjugate is attached to a gold electrode via a thiol-gold bond at the other end. The redox reporter molecule used the most is methylene blue. In general, production for E-DNA and E-AB sensors can take 12 to 48 working hours.
Methylene blue (Methylenblau) is a synthetic phenothiazine dye with redox properties first synthesized in 1876 by Heinrich Caro and tested by Paul Ehrlich in human patients. Ehrlich was able to demonstrate that it is useful for the treatment of malaria.
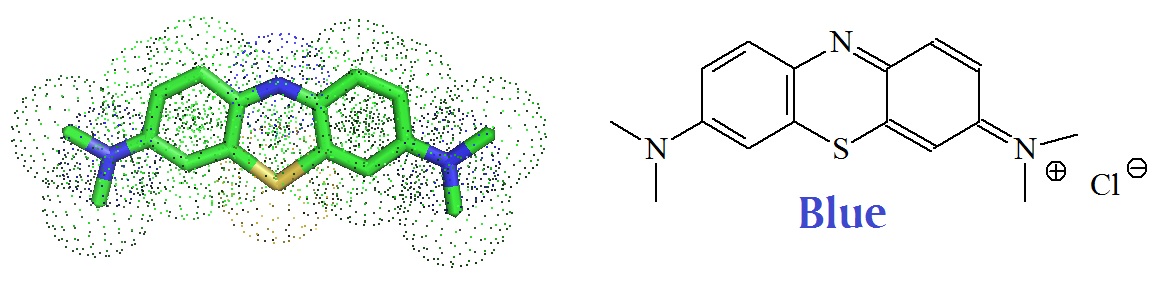
Methylene blue (MB): C16H18ClN3S, 319.851 g/mol [Pubchem].
“Methylene blue anhydrous is a compound consisting of dark green crystals or crystalline powder, having a bronze-like luster. Solutions in water or alcohol have a deep blue color. Methylene blue is used as a bacteriologic stain and as an indicator. It inhibits GUANYLATE CYCLASE, and has been used to treat cyanide poisoning and to lower levels of METHEMOGLOBIN.” [Source: MeSH].
“Methylene Blue is a synthetic basic dye. Methylene blue stains to negatively charged cell components like nucleic acids; when administered in the lymphatic bed of a tumor during oncologic surgery, methylene blue may stain lymph nodes draining from the tumor, thereby aiding in the visual localization of tumor sentinel lymph nodes. When administered intravenously in low doses, this agent may convert methemoglobin to hemoglobin.” [Source: NCIt].
Also, methylene blue was found to be useful as a therapeutic dye having antimicrobial activity, allowing staining of living tissues, found to be useful in diagnostic histopathology, has blood staining activities, is used as a medicinal photosensitizer, for cancer chemotherapeutic uses and psychoactive uses in dementia and psychosis. Critical clinical applications of methylene blues include the therapy of methemoglobinemia, septic shock, encephalopathy, and ischemia. Methylene blue as a photosensitizer, a stain, and a redox indicator has many other applications as well. Methylene blue intercalates with the DNA duplex.
Methylene blue’s autoxidizable chemical properties may be responsible for its unique biological action as both a metabolic energy enhancer and antioxidant that is frequently characterized by a biphasic dose-response.
The blue colored oxidized form of methylene blue (MB) can be reduced to colorless Leuco-Methylene Blue (LMB). Methylene blue is an autoxidizing dye that at low concentrations is in equilibrium with leuko-methylene blue forming a reversible redox-oxidation system. Methylene blue in its oxidized form can accept electrons from an electron donor. The colorless reduced form, leuko-methylene blue, acts as an electron donor. The transfer of electrons to oxygen forms water.
Methylene blue is also used as a photosensitizer for the production of singlet oxygen or in photodynamic therapy for the treatment of cancer. Methylene Blue is widely used as a redox indicator in analytical chemistry. A redox indicator undergoes a color change at a specific electrode potential.
Methylene blue can be covalently attached to DNA using flexible C12 or Cn alkyl linker units. The final construct is a sensitive redox reporter for DNA based electrochemical measurements. As reported by Pheeney and Barton (2012), the reduction of methylene blue conjugated to a DNA duplex occurs by two mechanisms on a DNA-modified electrode:
(1) Intercalated methylene blue tethered to the oligonucleotide duplex can be reduced in the DNA base pair stack, and
(2) by direct surface reduction at the electrode.
An example of an E-DNA sensor containing a stem-loop DNA modified on one terminal end with the redox reporter molecule and chemi-absorbed at the other end to a gold electrode is illustrated below.
The hybridization of a complementary target DNA to the stem region brakes the stem and the beacon linearizes. The linearization of the oligonucleotide reduces the electron transfer efficiency and decreases the observed current. Regeneration of the sensor is possible by washing with ultrapure water at room temperature.
Reference
Buchholz, K., Schirmer, R. H., Eubel, J. K., Akoachere, M. B., Dandekar, T., Becker, K., & Gromer, S. (2008). Interactions of Methylene Blue with Human Disulfide Reductases and Their Orthologues from Plasmodium falciparum . Antimicrobial Agents and Chemotherapy, 52(1), 183–191. http://doi.org/10.1128/AAC.00773-07.
Ferguson BS, Hoggarth DA, et al. (2013) Real-time, aptamer-based tracking of circulating therapeutic agents in living animals. Sci Transl Med, 5(213):213ra165.
Garrido, E., Pla, L., Lozano‐Torres, B., El Sayed, S., Martínez‐Máñez, R., & Sancenón, F. (2018). Chromogenic and Fluorogenic Probes for the Detection of Illicit Drugs. ChemistryOpen, 7(5), 401–428. http://doi.org/10.1002/open.201800034.
Liu Y, Tuleouva N, et al. (2010) Aptamer-based electrochemical biosensor for interferon gamma detection. Anal Chem, 82(19):8131–8136.
Muren, N. B., & Barton, J. K. (2013). Electrochemical Assay for the Signal-on Detection of Human DNA Methyltransferase Activity. Journal of the American Chemical Society, 135(44), 16632–16640. http://doi.org/10.1021/ja4085918
Oz, M., Lorke, D. E., Hasan, M., & Petroianu, G. A. (2011). Cellular and Molecular Actions of Methylene Blue in the Nervous System. Medicinal Research Reviews, 31(1), 93–117. http://doi.org/10.1002/med.20177
http://www.glenresearch.com/GlenReports/GR30-15.html
Sun, H., Zhu, X., Lu, P. Y., Rosato, R. R., Tan, W., & Zu, Y. (2014). Oligonucleotide Aptamers: New Tools for Targeted Cancer Therapy. Molecular Therapy. Nucleic Acids, 3(8), e182–. http://doi.org/10.1038/mtna.2014.32.
Wainwright M, Crossley KB. Methylene Blue-a therapeutic dye for all seasons? J Chemother. 2002;14:431–443. [PubMed]
Xiao Y, Lai RY, Plaxco KW. (2007) Preparation of electrode-immobilized, redox-modified oligonucleotides for electrochemical DNA and aptamer-based sensing. Nat Protoc, 2(11):2875–2880.
Zhu, C., Yang, G., Li, H., Du, D., & Lin, Y. (2015). Electrochemical Sensors and Biosensors Based on Nanomaterials and Nanostructures. Analytical Chemistry, 87(1), 230–249. http://doi.org/10.1021/ac5039863
---...---