GLP, GMP, cGMP, ISO 9001 and ISO 13485
Business technologies usually adhere to a set of standards and best practices. International organizations such as the International Organization for Standardization (ISO), the Industry Standards and Technology Organization (ISTO), the Institute of Electrical and Electronics Engineers (IEEE), or manufacturers of products publish guidelines for best practices used by various business technologies.
Best Practices
Best practices are methods or technics generally accepted as superior to alternatives. Best practices produce better results than those achieved by other means. Best practices maintain quality and are a feature of accredited management standards such as ISO 9000 and ISO 14001 family. [Best Practice].
GLP
Good Laboratory Practice (GLP) is a quality system concerned with organizational processes and conditions for planning health and environmental safety studies, design, performance, monitoring, recording, achieving, and reporting.
To ensure uniformity, consistency, reliability, quality, and the integrity of experimental results and products, research laboratories, and organizations developed quality systems of management controls. One of these is good laboratory practice or GLP. GLP applies to non-clinical studies conducted for the assessment of the safety of products in development. Also, the Organization for Economic Co-operation and Development (OECD) developed internationally accepted guidelines for the testing of chemicals. [GLP; OECD; Testing].
GMP
Good manufacturing practices (GMP) are guidelines recommended by agencies for practices controlling the authorization and licensing of manufacture and sale of food and beverages, cosmetics, pharmaceutical products, dietary supplements, and medical devices. These guidelines describe minimal requirements a manufacturer must meet to ensure that their products are consistently high.
The main goal of GMP is to prevent harm to the end-user. The main principles of GMP ensure that the end product is free from contamination; the manufacture is consistent and well documented; all personnel is well trained and that the product has been checked for quality more than just at the end phase. A well-designed quality management system ensures this. [GMP].
cGMP
Current good manufacturing practice (cGMP) refers to the Current Good Manufacturing Practice regulations enforced by the FDA. cGMP provides systems that assure proper design, monitoring, and control of manufacturing processes and facilities. Following cGMP guidelines ensures the identity, strength, quality, and purity of drug products. [cGMP facts; Regulatory Info].
Quality Management System (QMS)
A quality management system or QMS focuses on consistently meeting customer requirements and enhancing their satisfaction. QMS is a formalized system documenting processes, procedures, and responsibilities for archiving quality policies and objectives. A well designed QMS coordinates activities of an organization to meet customer and regulatory requirements. Also, QMS continuously improves an organization’s effectiveness. The international standard specifying requirements for a QMS is ISO-9001:2015. [QMS].
ISO = International Organization for Standardization
ISO 9001:2015
ISO 9001 refers to the international standard for a quality management system (QMS), the most noted standard concerning quality management. The ISO certification creates trust between customers, partners, and suppliers. Also, to get recertified, a company is continuously asked to improve. Continuous improvements assure that customers receive products and services that meet their requirements delivered consistently. ISO 9001 ensures that customers’ needs are satisfied and that internally, an organization profits from increased job satisfaction, improved morale, and improved operational results.
The ISO 9001 Standard sets forth the requirements for a company to follow, based on several quality management principles. These principles include a strong focus on customers, leadership motivation, and implication of top management, process-based approach, system approach to management, factual approach to decision making, and continual improvement. ISO 9001 ensures that customers receive consistent, high-quality products and services.
The QMS defines planning, control, assurance, and improvement activities to direct and control an organization concerning quality. An ISO-based QMS creates confidence in processes and products, providing the basis for continual improvement that ultimately leads to customer satisfaction and mutual success.
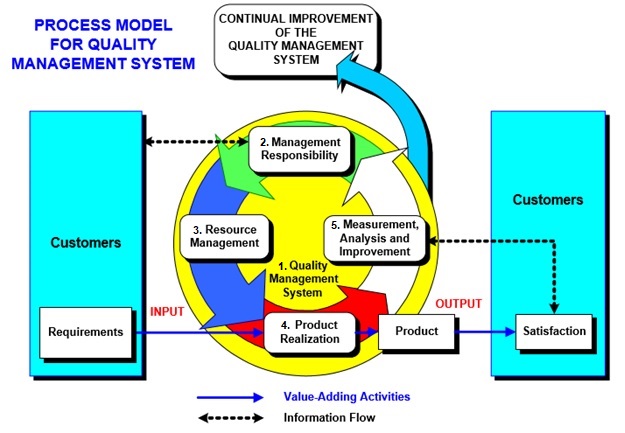
Figure 1: Example of the ISO 9001 QMS model. The ISO 9001 model is structured in five (5) major sections: 1. Quality Management System 2. Management Responsibility 3. Resource Management 4. Product Realization 5. Measurement, Analysis and Improvement.
The ISO 9001:2015 based QMS helps with the overall improvement and performance of an organization. When integrated, the QMS becomes a sustainable part of the company. Following the standard, organizations demonstrate their ability to consistently provide products and services that meet customer and regulatory requirements. Continuous improvements in products and services are also part of the standard. Implementation of a quality management system (QMS) ensures an organization’s sustained success. Further, the QMS increases the confidence of the organization or company in their ability to provide products and services consistently.
ISO 9001 requires certification. The current version is ISO 9001:2015. The “ISO 9001 Certificate” indicates that an organization has met the requirements in ISO 9001 that define the ISO 9001 Quality Management System (QMS). ISO 9001 evaluates and verifies if the QMS used is appropriate and effective, and the organization or company also identifies and implements improvements.
ISO 9001:2015 certification requires control of documents, control of records, internal audits, monitoring of non-conforming products and services, corrective actions, and preventive actions. Internal audits performed by the organization checks that its quality management system is working. Organizations can decide to invite an independent certification body for verifying their conformity to the standard. Alternatively, a client audit of the quality system is possible as well.
BSI ISO 9001:2016 Certificate
ISO 13485
ISO 13485, as an internationally agreed standard, defines requirements for a quality management system specific to the medical devices industry. ISO 13485 is modeled on ISO 9001 but focuses on components and products included in a finished medical device.
The ISO 13485 international standard focuses on meeting customer requirements, regulatory requirements, and maintaining the effectiveness of the QMS. ISO 13485 differs from ISO 9001:2015 by concentrating on meeting customer requirements and the maintenance of QMS effectiveness. ISO 13485 demonstrates to cusomers that quality systems are properly implemented and maintained. Furthermore, the ISO 13485 international standard requires additional documented procedures.
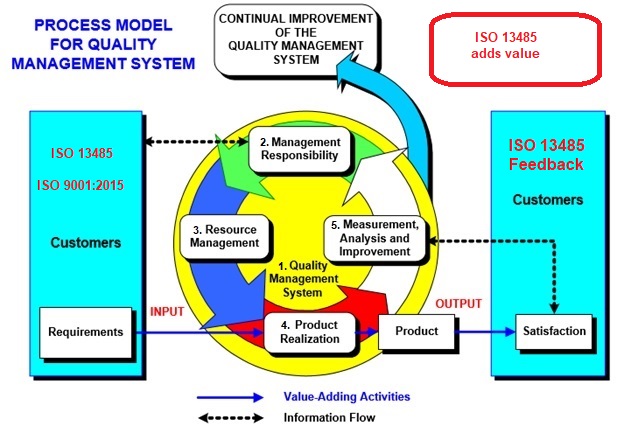
Figure 2: Example of the ISO 9001:2015-ISO 13485 QMS model. ISO 13485 adds value to the ISO 9001 QMS system.
Critical requirements of ISO 13485 are
- Risk management at all stages during product development and production.
- Strict design and development.
- Training and supervision of staff
- Prevention of contamination.
- Strict documentation and records requirements.
- Monitoring if customer requirements are met.
- Maintaining continued suitability and effectiveness of the QMS.
A third party acting as an auditor confirms if the standards are met. A certificate is issued after a successful external audit.
BSI ISO 13485:2016 Certificate
---...---