Inducing a deliberate immune response with vaccine adjuvants
Despite all of the recent advances in innate immunity and immune modulators or vaccine adjuvants, the induction of an immune response remains by a long way an empirical issue. While many vaccine adjuvants have been elucidated structurally and mechanistically, the common practice is to divide these compounds in two groups, pro-inflammatory Th1 and anti-inflammatory Th2 vaccine adjuvants. Yet, Th1-only adjuvants do not exist, as they always induce concomitant Th1 and Th2 immunities, however, some vaccine adjuvants may induce either a stronger or weaker Th2 immunity. A review of the known Th1/Th2 adjuvants, shows that the induced immunoresponse is different from adjuvant to adjuvant and that therefore we cannot assume that the immune response induced by complete Freund’s adjuvant (CFA) is the same as that induced by saponins like QS-21 or Quil A. Indeed, immunization using CFA in some cases rather than protecting against a pathogen potentiates infection, while immunization with Quil A results in a protection; despite the fact that both are Th1/Th2 adjuvants.
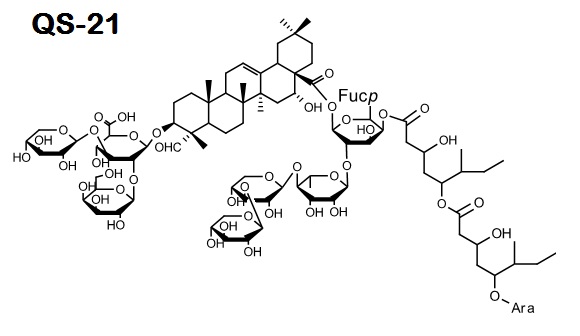
Figure 1: QS-21. Molecular formula: C92H148O46. Molecular weight: 1,990.13192 g/ml. QS-21 is an investigational adjuvant. this is a substance added to vaccines and other immunotherapies that is designed to enhance the body’s immune response to the antigen contained within the treatment. This vaccine additive is currently being evaluated in clinical trials.
Hence, while use of “generic” vaccine adjuvants, like CFA, to elicit antibody production for research purposes may be fine, that may not be the case when the goal is to develop a protective immune response against a pathogen or malignant cell, as in the case of vaccine development. Hence, in vaccine development, using CFA as the gold standard for immunity may lead to the wrong conclusions, i.e. an immune potentiating response against a pathogen, which may be protective when using a different vaccine adjuvant. Moreover, that different adjuvants that are ligands for toll-like-receptors (TLRs) of innate immunity show either synergistic or antagonistic effects, confirms the fact that these ligands are not all created equally, but that are quite different both structurally and functionally, a situation that may be used to modulate in a specific way immunity. Perhaps, it should be indicated that CFA’s adjuvanticity is due to a mixture of innate immunity ligands derived from Mycobacterium tuberculosis, suspended in an oil/water emulsion, yet as indicated above many of these ligands can be agonistic or antagonistic, which may explain some of the results obtained with CFA.
While there is a widespread assumption that the vaccine adjuvants’ mechanisms are still a mystery, the reality is very different. Since the discovery of innate immunity and saponin adjuvants the structure-function of these modulators has been well established. Indeed, we know their structures, receptors and in most instance the pharmacophores responsible for their immunomodulatory activity. Thus, in the case of vaccine development it is feasible to select the appropriate adjuvants to obtain a desired immune response. Moreover, we should realize that in inducing an immune response, the vaccine adjuvant is always in the driver’s seat particularly with protein antigens. Indeed, regardless of the antigen(s), adjuvants induce a systemic immunity that spreads through the body by means of cytokines and other immunological mediators; a result of the fact that an immunoresponse is largely caused by an infection, which seldom are localized in one part of the body. Yet, there have been attempts to use Th1/Th2 vaccine adjuvants in combination with peptide antigens lacking T-cell epitopes, with the expectation that the response would be an anti-inflammatory one; an assumption that ignores the adjuvant’s capacity to induce a systemic immunity that will take place despite the fact that the co-administered antigen lacks T-cell epitopes. Due to evolutionary reasons, adjuvants mainly induce an inflammatory Th1/Th2 immunity, which is the right response to identify and destroy foreign pathogens as well as intra-cellularly infected and malignant cells.
However, in some cases like in proteinopathies, e.g. Alzheimer’s disease, multiple sclerosis (MS) and others, a Th1/Th2 inflammatory response is damaging, destroying organs in an irreversible way. In such a case, the desired immunity would be an anti-inflammatory one Th2-only. But, in contrast to vaccine adjuvants inducing Th1/Th2 immunity, which are common, those that induce Th2-only are quite rare; indeed the only one being used now is alum. But alum is a poor vaccine adjuvant in the very young and the elderly, as shown by the poor efficacy of the flu vaccine in the older population, i.e. while the efficacy is near 90 percent in the population around 20 years old, it decreases to 40 percent or lower in those older than 65. The reasons for the poor availability of Th2 adjuvants are evolutionary; while an inflammatory immunity designed to kill foreign invaders was developed quite early by multi-cellular organisms, the anti-inflammatory immunity was developed hundreds of millions of years after the inflammatory immunity. Indeed, these anti-inflammatory Th2 modulators appeared when parasites, besides unicellular become multi-cellular or metazoan parasites like helminths; i.e. destruction of these parasites will cause damage to both the parasite and the host. Hence, the milder humoral Th2 immunity appeared to help to heal the damage caused by the parasites, this way establishing an immunity that minimized damage to the host and the parasite.
Evolutionarily, the metazoan parasites developed compounds that mimic those produced by mammals to prevent rejection of the fetus, indeed agents that inhibited the inflammatory response while inducing an anti-inflammatory response. Their recent development, less than 400 million years would explain why these anti-inflammatory agents are not so common as compared to those for innate immunity. In fact, those compounds are based on 2 pharmacophores, phosphorylcholine (PC) and fucose. The PC is responsible for the immune modulatory effects of sphingosine-1-phosphate (S1P), which is produced by phosphorylation of sphingolipids by two sphingoside kinases and it is essential for immune-cell trafficking; i.e. S1P seems to favor Th2 and Th17, while dampening Th1 immunoresponses. In contrast to PC, fucosylated glycans act at the antigen presenting cells (APCs) level, i.e. dendritic cells (DCs) and macrophages, via the cell lectin DC-SIGN, which is also a receptor for HIV, Ebola and other viruses. In fact, DC-SIGN can accept to kinds of different ligands, those carrying either mannose or fucose, yet, the induced immune response is different with each ligand. Upon binding to the lectin, mannose ligands bias the immune response toward a pro-inflammatory one, while fucose ligands bias it toward an anti-inflammatory response. Hence, induction of Th2 immunity will require alum, a poor adjuvant, or vaccine adjuvants with either PC or fucose.
From the practical point of view, while CFA can be used to stimulate Th1 immunity with a useful antibody production for research purposes, such a response would not be satisfactory for vaccine purposes where the immune response needs to be more define. Indeed, preliminary vaccine studies with CFA may deliver misleading results that cannot be extended to other Th1 adjuvants. Thus, in vaccine development new vaccine adjuvants, e.g. TLR ligands should be considered (Table 1). An advantage of these vaccine adjuvants is that they can show a synergistic effect, which results in an immune response that it is much better than that the sum of the responses induced by the separate adjuvants. Yet, some of these adjuvants may also show antagonistic effects on the immune response.
Table 1. Properties of some vaccine adjuvants
a Defines the stimulated immunity.
b Toll Like Receptor
c These adjuvants belong to the Quillaja saponins group.
d MPL: monophosphoryl lipid A
e CpG DNA
f double stranded RNA
N/A: Non applicable
Reference
Rajput, Z. I., Hu, S., Xiao, C., & Arijo, A. G. (2007). Adjuvant effects of saponins on animal immune responses . Journal of Zhejiang University. Science. B, 8(3), 153–161. http://doi.org/10.1631/jzus.2007.B0153