The spike (S) protein of the virus mediates receptor binding and membrane fusion. The sequence specificity also defines the range of the hosts and the specificity of the virus. Also, the S protein is the target for a variety of neutralizing antibodies and vaccine design.
Gene recombination or mutations of the receptor-binding domain (RBD) appears to allow transmission between different hosts. Some of these mutations can lead to a higher mortality rate of the infected hosts. Some observed mutations are in the reference sequence for the spike protein. Korber et al. developed a analysis pipeline to track mutations in the SARS-CoV-2 spike protein. However, the clinical evidence of all these mutations still needs to be verified.
Schematic of the coronavirus spike (S) protein domain structure
.jpg)
The location of receptor-binding subunit S1, the membrane-fusion subunit S2, the transmembrane anchor (TM), the intercellular tail (IC) are indicated. The location of the S1 N-terminal domain (S1-NTD), the S1 C-terminal domain (S1-CTD), the fusion peptide (FP), and the heptade repeat regions N and C (HR-N and HR-C) are indicated as well.
The following image shows the annotated sequence for SARS-CoV-2 spike protein mutations from the coronavirus associated with COVID-19 originating in Wuhan of Hubei province in China. Source: ORIGIN YP_009724390, 1273 a, surface glycoprotein [Severe acute respiratory syndrome coronavirus 2].
A new SARS-CoV-2 virus emerged in the UK containing a noval set of spike mutations named B.1.1.7. This new virus strain appears to be more infectious.
The potential biological effects of these mutations are:
(i) Mutation N501Y is part of six key contact residues within the receptor-binding domain (RBD) thought to increase binding
affinity to human and murine receptor protein ACE2.
(ii) The spike deletion 69-70del may help the virus to evade the human immune response.
(iii) Mutation P681H is close to the furin cleavage site.
Other identified mutations in B.1.1.7 spike protein are: A570D, T716I, S982A, D1118H.
Wibmer et al., and others, recently, January 2021, described the emergence of a new SARS-CoV-2 lineage in South Africa, 501Y.V2.
Nine changes in the spike protein were found. One cluster in the N-terminal domian (NTD) includes four substitutions and a deletion (L18F, D80A, D215G, Δ242-244, and R246I). Another cluster contained three substitutions in RBD (K417N, E484K, and N501Y). The 501Y change has been associated with increased transmissibility, rather than immune pressure. The accumulation of these mutations may possibley allow the virus to escape from neutralization by earleir aquired antibodies. Mutations in the same regions have also been described in a new variant, which has recently emerged independently in Brazil.
Omicron Variant B.1.1.529
More recently, on November 24, 2021, a new SARS-CoV-2 variant, B.1.1.529 was identifed in South Africa. This variant is called the Omicron variant. The spike protein of this variant has at least 30 amino acid substitutions, three small deletions, and one small insertion. Fifteen (15) of the 30 amino acid substituions are located in the receptor binding domain.
Key Amino Acid Substitutions in Spike Protein:
A67V, del69-70, T95I, del142-144, Y145D, del211, L212I, ins214EPE, G339D, S371L, S373P, S375F, K417N, N440K, G446S, S477N, T478K, E484A, Q493R, G496S, Q498R, N501Y, Y505H, T547K, D614G, H655Y, N679K, P681H, N764K, D796Y, N856K, Q954H, N969K, L981F
N-term. S1
1 MFVFLVLLPL VSSQCVNLTT RTQLPPAYTN SFTRGVYYPD KVFRSSVLHS TQDLFLPFFS S1
---------- -------F-- ---------- ---------- ---------- ----------
SA501Y.V2 L18F
61 NVTWFHAIHV SGTNGTKRFD NPVLPFNDGV YFASTEKSNI IRGWIFGTTL DSKTQSLLIV
--------|| ---------Y ---------- -------F-- ---------- ----------
---------- ---------A[SA 501Y.V2 D80A]----- ---------- ----------
B.1.1.7 Del S:D80Y S:S98F
------V-|| ---------- ---------- ----I----- ---------- ----------
Omicron B.1.1.529 Deletion
121 NNATNVVIKV CEFQFCNDPF LGVYYHKNNK SWMESEFRVY SSANNCTFEY VSQPFLMDLE
---------- ---------- ---|------ ---------- ---------- ----------
B.1.1.7 Deletion
---------- ---------- -|||D----- ---------- ---------- ----------
Omicron B.1.1.529 Deletion
181 GKQGNFKNLR EFVFKNIDGY FKIYSKHTPI NLVRDLPQGF SALEPLVDLP IGINITRFQT
---------- ---------- ---------- ----G----- -V-------- ----------
SA501Y.V2 D215G 20A.EU1
---------- ---------- ---------- |--------- ---------- ----------
EPE insertion 214
241 LLALHRSYLT PGDSSSGWTA GAAAYYVGYL QPRTFLLKYN ENGTITDAVD CALDPLSETK
---------- ---------- ---------- ---------- ---------- ----------
-[.]-I----
DELETION[SA501Y.V2 242-244; R246I]
301 CTLKSFTVEK GIYQTSNFRV QPTESIVRFP NITNLCPFGE VFNATRFASV YAWNRKRISN RBD
---------- ---------- ---------- ------SL-- IL-S---S-- -S-D----N-
---------- ---------- ---------- --------D- ---------- ----------
361 CVADYSVLYN SASFSTFKCY GVSPTKLNDL CFTNVYADSF VIRGDEVRQI APGQTGKIAD
------F--S -T----LN-- ---LA----- --P-I----- ----VQ-IE- ---R--N---
---------- -S-----R-- ---------- ---------- ---------- ---E------
---------- ---------- ---------- ---------- ---------- ---A---N--
[SA501Y.V2 K417N]
---------- L-P-F----- ---------- ---------- ---------- ------N---
421 YNYKLPDDFT GCVIAWNSNN LDSKVGGNYN YLYRLFRKSN LKPFERDIST EIYQAGSTPC
---------- ----SR--KK ---RFV---- -RF--L-R-- ---------- QV---SNIS-
20A.EU2
---------- ---------- -----S---- -----E-Q-- ---------- ------R---
---------- ---------- ---------- ---------- ---------- ------I---
---------- ---------K -----S---- ---------- ---------- ------NK--
481 NGVEGFNCYF PLQSYGFQPT NGVGYQPYRV VVLSFELLHA PATVCGPKKS TNLVKNKCVN
DSA-SS---S ---P----R- Y-F-C--H-A PA-------- ---------- ----------
--IK------ ---------- Y--------S RS-------- ---------- ----------
[SA501Y.V2 E484K] B.1.1.7[SA501Y.V2 N501Y]
---A------ --R--S-R-- Y---H----- ---------- ---------- ----------
Region of transmissibility ?
541 FNFNGLTGTG VLTESNKKFL PFQQFGRDIA DTTDAVRDPQ TLEILDITPC SFGGVSVITP
---------- ---------- ---------D ---------- ---------- ----------
B.1.1.7
------K--- ---------- ---------- ---------- ---------- ----------
601 GTNTSNQVAV LYQDVNCTEV PVAIHADQLT PTWRVYSTGS NVFQTRAGCL IGAEHVNNSY
---------- ---G------ ---------- ---------- ---------- ----------
D614G
---------- ---G------ ---------- ---------- ---------- ----Y-----
FURIN 1 2
.......................<-S1|S2->.....<-|->.........................
661 ECDIPIGAGI CASYQTQTNS PRRAR|SVASQ SIIAY|TMSLG AENSVAYSNN SIAIPTNFTI
---------- ---------- H-A-A ----- ----- ----- ---------- -----I----
B.1.1.7 B.1.1.7
---------- --------K- H--------- ---------- ---------- ----------
721 SVTTEILPVS MTKTSVDCTM YICGDSTECS NLLLQYGSFC TQLNRALTGI AVEQDKNTQE
---------- ---------- ---------- ---------- ---K------ ----------
S2’TMPRSS2
........INTERNAL FUSION PEPTIDE.....<-|->.....FUSION PEPTIDE.....
781 VFAQVKQIYK TPPIKDFGGF NFSQILPDPS KPSKR|SFIED LLFNKVTLAD AGFIKQYGDC
---------- -----Y---- ---------- ------ P--- ---------- ----------
841 LGDIAARDLI CAQKFNGLTV LPPLLTDEMI AQYTSALLAG TITSGWTFGA GAALQIPFAM
---------- -----K---- ---------- ---------- ---------- -P------P-
901 QMAYRFNGIG VTQNVLYENQ KLIANQFNSA IGKIQDSLSS TASALGKLQD VVNQNAQALN HR1
---------- ---------- ---------- ---------- -P-------- ---H------
961 TLVKQLSSNF GAISSVLNDI LSRLDKVEAE VQIDRLITGR LQSLQTYVTQ QLIRAAEIRA
--------K- ---------- FA---PP--- ---------- ---------- ----------
B.1.1.7
1021 SANLAATKMS ECVLGQSKRV DFCGKGYHLM SFPQSAPHGV VFLHVTYVPA QEKNFTTAPA
---------- ---------- ---------- ---------- ---------- ----------
1081 ICHDGKAHFP REGVFVSNGT HWFVTQRNFY EPQIITTDNT FVSGNCDVVI GIVNNTVYDP
---------- ---------- ---------- -------H-- ---------- ----------
B.1.1.7
1141 LQPELDSFKE ELDKYFKNHT SPDVDLGDIS GINASVVNIQ KEIDRLNEVA KNLNESLIDL HR2
1201 QELGKYEQYI KWPWYIWLGF IAGLIAIVMV TIMLCCMTSC CSCLKGCCSC GSCCKFDEDD CPD
1261 SEPVLKGVKL HYT
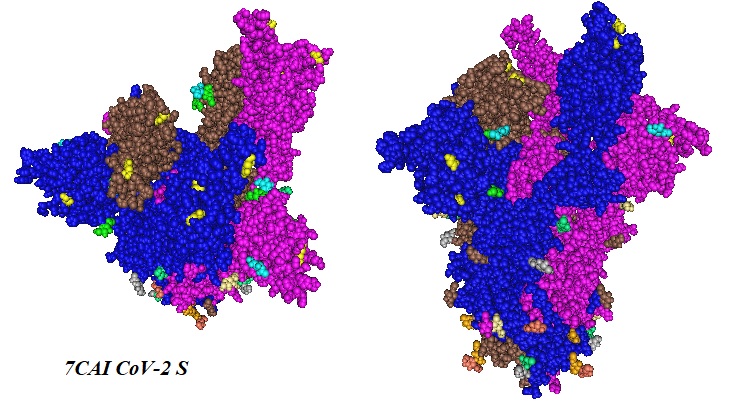
Figure 1: Location of observed mutations for SARS-CoV-2 lineage, South Africa, 501Y.V2 are shown in yellow in the trimeric S protein structure published by Deng et al. in 2020 (PDB ID 7CAI). Residues shown in different colors depict glycosylation sites.
Reference
Antiviral Peptides SARS-CoVs
B.1.1.7 : Preliminary genomic characterisation of an emergent SARS-CoV-2 lineage in the UK defined by a novel set of spike mutations
Report written by: Andrew Rambaut, Nick Loman, Oliver Pybus, Wendy Barclay, Jeff Barrett, Alesandro Carabelli, Tom Connor, Tom Peacock, David L Robertson, Erik Volz, on behalf of COVID-19 Genomics Consortium UK (CoG-UK).
Coutard B, Valle C, de Lamballerie X, Canard B, Seidah NG, Decroly E. The spike glycoprotein of the new coronavirus 2019-nCoV contains a furin-like cleavage site absent in CoV of the same clade. Antiviral Res. 2020 Apr;176:104742. [PMC]
Coronavirus Genomes
COVID Vaccine Peptides
Lv Z, Deng YQ, Ye Q, Cao L, Sun CY, Fan C, Huang W, Sun S, Sun Y, Zhu L, Chen Q, Wang N, Nie J, Cui Z, Zhu D, Shaw N, Li XF, Li Q, Xie L, Wang Y, Rao Z, Qin CF, Wang X. Structural basis for neutralization of SARS-CoV-2 and SARS-CoV by a potent therapeutic antibody. Science. 2020 Sep 18;369(6510):1505-1509. [PMC]
Du L, He Y, Zhou Y, et al. The spike protein of SARS-CoV--a target for vaccine and therapeutic development. Nature reviews. Microbiology. 2009 Mar;7(3):226-236. [PMC]
Emma B. Hodcroft, Moira Zuber, Sarah Nadeau, Iñaki Comas, Fernando González Candelas, SeqCOVID-SPAIN consortium, Tanja Stadler, Richard A. Neher; Emergence and spread of a SARS-CoV-2 variant through Europe in the summer of 2020. medRxiv 2020.10.25.20219063.
Isabel, S., Graña-Miraglia, L., Gutierrez, J.M. et al. Evolutionary and structural analyses of SARS-CoV-2 D614G spike protein mutation now documented worldwide. Sci Rep 10, 14031 (2020). [pdf]
B Korber, WM Fischer, S Gnanakaran, H Yoon, J Theiler, W Abfalterer, B Foley, EE Giorgi, T Bhattacharya, MD Parker, DG Partridge, CM Evans, TM Freeman, TI de Silva, on behalf of the Sheffield COVID-19 Genomics Group, CC LaBranche, DC Montefiori; Spike mutation pipeline reveals the emergence of a more transmissible form of SARS-CoV-2. bioRxiv 2020.04.29.069054. [pdf]
Lai AL, Millet JK, Daniel S, Freed JH, Whittaker GR. The SARS-CoV Fusion Peptide Forms an Extended Bipartite Fusion Platform that Perturbs Membrane Order in a Calcium-Dependent Manner. J Mol Biol. 2017 Dec 8;429(24):3875-3892. [PMC]
Shen S, Tan TH, Tan YJ. Expression, glycosylation, and modification of the spike (S) glycoprotein of SARS CoV. Methods Mol Biol. 2007;379:127-35. [PMC]
Structure and sequence of SARS-CoV-2
Tang T, Bidon M, Jaimes JA, Whittaker GR, Daniel S. Coronavirus membrane fusion mechanism offers a potential target for antiviral development. Antiviral Res. 2020 Jun;178:104792. doi: 10.1016/j.antiviral.2020.104792. [PMC]
Tegally et al. 2021; Detection of a SARS-CoV-2 variant of concern in South Africa. Nature 592, 15 April 2021, 438 - 443. [Nature]
Therapeutic Peptides
Therapeutic strageties for vaccination
Wibmer et al. 2021. SARS-CoV-2 501Y.V2 escapes neutralization by South African COVID-19 donor plasma bioRxiv preprint doi: https://doi.org/10.1101/2021.01.18.427166.
Xiao X, Dimitrov DS. The SARS-CoV S glycoprotein. Cell Mol Life Sci. 2004 Oct;61(19-20):2428-30. [PMC]
---...---