In RNA interference, small interfering RNAs recognize messenger RNA homologs in cells and induce their degradation.
When developing siRNA-based drugs for therapeutic use, problems encountered are their low efficiency of delivery to targeted cells and degradation by cellular nucleases. A promising approach to improve cellular delivery of siRNA is bioconjugation. Like other oligonucleotides, siRNA conjugation to lipophilic molecules, antibodies, aptamers, ligands, peptides, or polymers enhances cellular delivery and selective targeting.
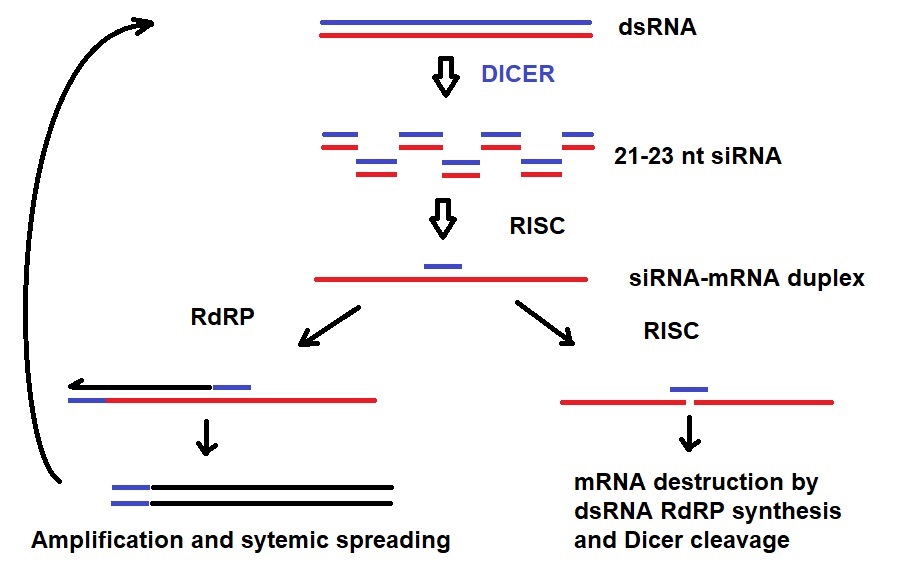
Figure 1: Mechanism of systemic and transitive RNAi. Dicer cleavage of dsRNA into 21-23 nt siRNA initiates RNAi. RISC unwinds siRNA duplexes. siRNA binds to mRNA. Either the targeted mRNA is destructed or amplified via RdRP. Amplified dsRNA can transitively silence a 5’-located mRNA in a second cell {Engelke, David R.: RNA interference (RNAi). Nuts and bolts of RNAi technology. DNA Press 2003}.
Table 1: Phosphate Backbone Modifications and their Effect.
|
Modification
|
Structure
|
ΔTm duplex per modification
|
Impact on the efficiency of RNAi
|
Others
|
|
Phosphorothioate (PS)
|
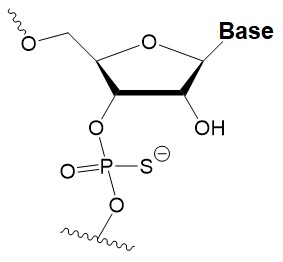
|
−0.7°C
|
PS inhibits RNAi when introduced in the central part of the antisense strand.
|
PS protects siRNAs from the action of exoribonucleases in vitro and in vivo.
>50% PS cause toxicity in vitro and in vivo.
|
|
Dimethylethylenediamine (DMEDA)
|
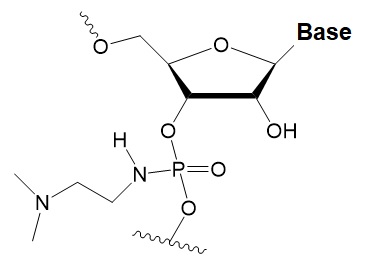
|
−0.7–3.4°C
(shown only for thymidine)
|
10% DMEDA in the sense strand are tolerated by RNAi.
|
The effect on nuclease resistance of siRNA is not known.
|
|
Tert-butyl-S-acyl-2-thioethyl (tBu-SATE)
|
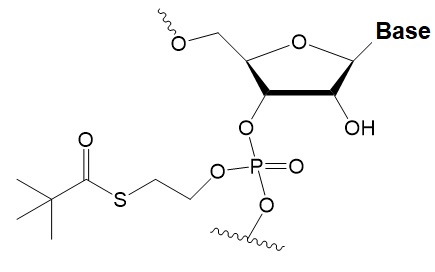
|
No data.
|
25% tBu-SATE are tolerated by RNAi.
|
≥20–40% tBu-SATE in siRNA increase nuclease resistance in vitro and in vivo.
Increases hydrophobicity of siRNA.
Cleaved by thioesterase in the cytoplasm of the cell giving a phosphodiester bond.
|
|
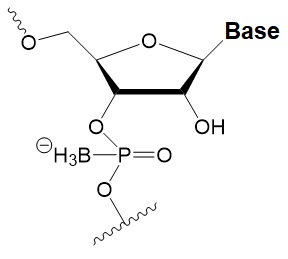
Boranophosphate (BP)
|
|
+0.4–1°C (<50% of siRNA)
−0.8–2.5°C (>50% of siRNA)
|
>50% PB inhibit RNAi, the central part of the antisense strand is the most sensitive to modifications.
|
Approximately two times more effectively protect against ribonucleases than PS, but do not cause toxicity in vitro.
|
|
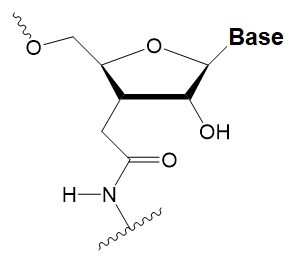
Amide linker
|
|
−0.3 to +0.9°C
|
In some siRNA positions, a single substitution for an amide linker is tolerated by RNAi.
|
The introduction of two amide linkers from the 3′ ends of the duplex increases the nuclease resistance of siRNA in serum.
|
| |
|
|
|
|
|
Adapted from: Chernikov IV, Vlassov VV, Chernolovskaya EL. Current Development of siRNA Bioconjugates: From Research to the Clinic. Front Pharmacol. 2019 Apr 26;10:444. doi: 10.3389/fphar.2019.00444. PMID: 31105570; PMCID: PMC6498891. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC6498891/.
---...---