Amino Acid Analysis / Testing refers to the quantitative analysis of either free amino acids or amino acids released from proteins via hydrolysis present in biological samples such as blood, plasma, urine, foods, dietary or health supplements, nutraceuticals, beverages, tablets, supplement capsules, plant extracts and any other samples or matrices that contain amino acids.
Proteins, peptides and amino acids are found in a great variety of food sources including animals, fungi, vegetables, cereals and many others. It has been shown by many authors that due to their complexity the identification of these compounds require the use of advanced analytical techniques. Many techniques including high-performance liquid chromatography, sometimes referred to as high-pressure liquid chromatography (HPLC), gas chromatography (GC), capillary electrophoresis (CE), nuclear magnetic resonance (NMR), Fourier transform infrared spectroscopy (FTIR), inductively coupled plasma mass spectrometry (ICP-MS), inmunosensors, and others have been used or were investigated for their use to analyze amino acids in different matrices in the past. Furthermore, amino acids have been identified and quantified in different natural matrices using micellar electrokinetic chromatography (MEKC), micro chip electrophoresis or HPLC. The US Pharmacopeia Dietary Supplement Chapter lists detailed test methods for each individual amino acid ingredient as follows: the identification of amino acids is perform by FTIR, analysis of chromatographic impurities is performed by a TLC–ninhydrin based method and the determination of the potency of raw materials is done by using titration assays. However, these are classical or older methods. The newer, more recent methods that were developed during the last decades utilize automated instruments called “amino acid analyzers.” Most amino acid analysis or testing labs are now capable of routinely testing amino acids as either the free amino acids or those released from protein hydrolysates.
Amino acid analysis enables the precise determination of protein quantities and provides detailed information regarding the relative amino acid composition of proteins and free amino acids present in physiological samples. The resulting amino acid composition is a characteristic profile for a protein. In some cases this profile allows to identify a protein via database searches. A typical amino acid analysis includes a hydrolysis step followed by separation, and detection and quantification of the released and labeled amino acids. Briefly, samples that contain proteins are subjected to acid hydrolysis and are broken down into individual amino acids. Next, the released amino acids are reacted with a derivatizing reagent, converted to stable chromophore modified amino acids and are separated and analyzed by HPLC.
Amino acids present in finished goods such as tablets, capsules, powders, and in liquid syrups can all be analyzed using automated amino acid analysis. The analysis of single and/or multiple amino acid preparations can be performed and the results are documented according to the specific guidelines.
What are amino acids?
Amino acids are the "building blocks" of proteins and are found in every human tissue as well as all mammalian tissues. Amino acids play major roles in nearly every chemical process in the human body. Amino acids are vital molecules that affect both physical and mental functions in mammals and humans.
Proteins are found in the brain, the nervous system, ligaments, tendons, bones, as well as in antibodies, and are vital for the regulation of enzymes and blood transport proteins. Only twenty amino acids of the naturally occurring amino acids are found in proteins because they are the only ones that are coded for by the genetic code.
Chemically, an amino acid is an organic acid in which one of the hydrogen atoms on a carbon atom has been replaced by an ammonia group (NH2). The term amino acid usually refers to an aminocarboxylic acid. One particular type of amino acids called α-amino acids are of the general formula R-CHNH2-COOH with the NH2 group in the alpha position. All amino acids, except the amino acid glycine, are stereoisomers and only the L-form or L-enantiomer, which is one mirror image, of the possible two amino acids, is biologically active in an organism. The L refers to L-stereoisomers or left-handed isomers of the amino acids. These L-forms are the result of the hydrolysis products of proteins. All proteins consist of groups of different types and percentages of L-amino acids. Sometimes, in rare instances, these classes of molecules also include α–amino phosphoric acids and α–aminosulfonic acids. The following structures illustrate the nature of stereoisomers.
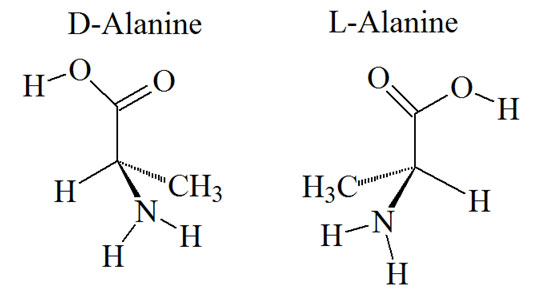 Stereoisomers of alanine
Stereoisomers of alanineThe models of D- and L-alanine are used here as an example to describe the nature of stereo isomers. The four bonds of the central alpha carbon of an amino acid point towards the four corners of a tetrahedron. Alanine has a methyl group (CH3) as a side chain group. If we imagine holding the model of the structure with the carboxyl group (COOH) at the top and the amino group (NH2) at the bottom, the CH3 group in the D form will be on the right, and in the mirror image L form will be on the left.
Amino acids are important for the function of an organism and are part of its metabolism. The misregulation of biochemical processes that involve amino acids in the human body can lead to metabolic diseases. An example is the presence of excessive amounts of specific amino acids in the blood called “aminoacidemia.” Another example is the excessive excretion of amino acids in the urine called “aminoaciduria.” In addition, an inherited disorder called hyperbasic aminoaciduria is associated with a deficiency of a dibasic amino acid transport. Individuals with this disorder do not display protein intolerance. Because of the great number of metabolic disorders that involve amino acids and the wide range of affected systems, nearly every "presented complaint" to a doctor may have a congenital metabolic disease as a possible cause. This is especially true in childhood. Therefore, it is safe to reason that amino acid analysis or testing is an important technique for the analysis of metabolites that contain primary and secondary amino groups in their structure, which are present in humans or mammals as well as in food stuff, nutraceuticals and health supplements. This type of analysis is also quite important in clinical chemistry.
Except for the nine so called "essential amino acids," the human body can synthesize all of the amino acids necessary for the biochemical synthesis of proteins. Plants and micro-organisms can synthesize all amino acids from small precursor molecules. These nine amino acids must be included in the human diet or taken as supplements to be available in adequate amounts. The failure to obtain enough of even one of these essential amino acids can result in serious health implications and the degradation of the body's proteins. These phenomena can lead to the destruction of muscle and other protein structures to obtain the amino acids that the body needs to survive. Because of this, amino acids are classified into two main groups: essential and non-essential amino acids. There are nine essential amino acids in the group: phenylalanine, valine, threonine, tryptophan, isoleucine, methionine, leucine, lysine, and histidine. In some biochemical and molecular biology books, arginine and histidine are sometimes also listed as essential amino acids. Non-essential amino acids are amino acids that can be produced by the human body. These include: alanine, aspartic acid, arginine, cysteine, cystine, glutamine, glycine, serine, tyrosine, hydroxyproline, asparagine, and proline. However, non-essential amino acids are also used as a supplement for cell culture medium to increase cell growth and cell viability. Furthermore, protein sources that have the full complement of essential amino acids are sometimes called complete proteins. Whereas proteins that are devoid or deficient in one or more essential amino acid are called incomplete proteins. Arginine is essential during times of rapid growth. It is particularly needed during childhood or when muscle growth is initiated during weight training sessions. Furthermore, the amounts of each essential amino acid required depends on the overall amino acid composition of the proteins that are consumed. For example, human cells can synthesize cysteine from methionine if needed. However, if the intake of cysteine is low extra methionine is needed in the diet not only to meet the needed methionine levels but also to synthesize cysteine.
For nutritional purposes, the protein quality can be estimated and a chemical score can be defined. Usually this is done by assigning a chemical score of one hundred (100) to whole egg protein and by comparing the proteins of interest to this protein. This score implies that the whole egg protein is completely utilized. The amino acid that gives the lowest score in relation to the reference protein will dictate the final chemical score. This amino acid is called the first limiting amino acid. The following formula is used to calculate the chemical score:
| Chemical score = |
[ amount of essential amino acid in test protein (mg/g)] |
| --------------------------------------------------------------------------------------------------------------------- |
| [ amount of same essential amino acid in reference protein or egg protein ] |
Proteinogenic amino acids are amino acids that are used by the cellular machinery of an organism to synthesize proteins. These amino acids are encoded for in the genetic code. There are 22 standard amino acids, but only 21 are found in eukaryotes. Selenocysteine and pyrrolysine are incorporated into proteins by different biosynthetic mechanisms. The other 20 are directly encoded by the universal genetic code.
Branched-chain amino acids, a term used by the food and supplement industry, are amino acids that contain branched aliphatic side-chains. The side chains contain a carbon atom that is connected to more than two other carbon atoms. There are three branched-chain amino acids: leucine, isoleucine and valine. The structures of these amino acids are shown below.
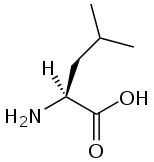 |
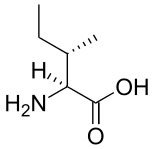 |
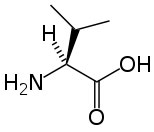 |
| Leucine |
Isoleucine |
Valine |
Branched-chain amino acids
In muscle proteins these three branched-chain amino acids account for circa 35% of the essential amino acids and 40% of the preformed amino acids required by mammals.
Amino acid profiles that determine the presence or absence and the quantities of the twenty amino acids in plasma can illuminate problems in amino acid status of a human patient. This is done by determining essential amino acid imbalances. Amino acid profiles containing up to 45 compounds can be even more informative. These evaluation types allow identifying the status of essential, branched chain, and other non-essential amino acids. Together, with the analysis of vitamins and minerals, this analysis allows identification of neuroendocrine, vascular, detoxification, and functional vitamin and mineral deficiencies.
Fasting plasma amino acid levels represent a homeostatic balance between supply and utilization of amino acids, making amino acid analysis of plasma amino acids ideal for repeated assessments by monitoring the progress of treatment. Collecting a fasting plasma specimen from a patient removes recent dietary intake effects. The following factors can reflect changes over time in plasma: chronic dietary intake, digestive efficiency, hepatic uptake and skeletal muscle's ability to maintain transamination. Amino acid analysis or testing helps monitoring amino acid levels involved in nutritional function, including gastrointestinal function, cellular energy production, detoxification, neurotransmitter metabolism, muscle catabolism, collagen synthesis and maintenance, as nutritional markers, and can be indicators of vascular function. Furthermore, amino acid analysis or testing of amino acid levels in plasma is useful for the design of supplement formulas. For example, to manufacture customized amino acid blends based on a patient's specific test results. The notion here is that these customized amino acid formulations will provide appropriate amounts of essential and conditionally essential amino acids that can be delivered in a balanced ratio to offset the risk of imbalance, sometimes observed when single amino acid supplements are used. Known conditions associated with amino acid changes in plasma include cardiovascular disease, depression, anxiety, insomnia, chronic fatigue syndrome, multiple sclerosis, rheumatoid arthritis, epilepsy, congestive heart failure, impotence and erectile pain syndromes, multiple chemical sensitivities, detoxification disorders, autism spectrum disorders, Alzheimer's disease, hypothyroidism, arrhythmias, hypertension, ADD/ADHD, and infertility. 10/31/2013
Table 1: The Genetically-Encoded Amino Acid Set
| 1 |
Alanine, Ala, A |
11 |
Methionine, Met, M |
| 2 |
Cysteine, Cys, C |
12 |
Asparagine, Asn, N |
| 3 |
Aspartic acid, Asp, D |
13 |
Proline, Pro, P |
| 4 |
Glutamic acid, Glu, E |
11 |
Glutamine, Gln, Q |
| 5 |
Phenylalanine, Phe, F |
15 |
Arginine, Arg, R |
| 6 |
Glycine, Gly, G |
11 |
Serine, Ser, S |
| 7 |
Histidine, His, H |
17 |
Threonine, Thr, T |
| 8 |
Isoleucine, Ile, I |
18 |
Valine, Val, V |
| 9 |
Lysine, Lys, K |
11 |
Tryptophan, Trp, W |
| 10 |
Leucine, Leu, L |
20 |
Tyrosine, Tyr, Y |
Table 2: Other Common Amino Acids in Proteins
| 1 |
Hydroxyproline (hydroxylated proline-two isomers) |
| 2 |
Cystine (oxidised cysteines) |
| 3 |
Pyroglutamic acid (cyclized N-terrninal glutamic acid) |
Table 3: Other Amino Acids Commonly used in Peptide Design
| 1 |
Alpha-amino butyric acid (cysteine replacement) |
| 2 |
Beta-amino alanine (straight chain isomer of alanine) |
| 3 |
Norleucine (linear sidechain isomer of leucine) |
Table 3: Grouping by hydrophilicity and hydrophobicity
| Hydrophilic |
D, E, H, K, N, Q, R, S, T, hydroxyproline, pyroglutamic acid |
| Hydrophobic |
A, F, I, L, M, P, V, W, Y, Alpha-amino butyric acid, beta-amino alanine, norleucine |
| Indeterminate |
C,G |
Table 3: Other Groupings of Amino Acids
| Amino acids subject to oxidation under relatively mild conditions |
cysteine, methionine |
| Amino acids subject to deamidation or dehydration |
asparagine, glutamine, C-terminal amides |
| Amino acids subject to degradation during peptide preparation |
methionine, tryptophan |
| Amino acids which can carry a positive charges |
lysine, arginine, N-terminal end of peptide, histidine |
| Amino acids which can carry a negative charge |
aspartic acid, glutamic acid, C-terminal end of peptide, tyrosine |