Bioconjugate Chemistry for Molecular Engineering
Recent developments in protein engineering, also known as bioengineering, have enabled the production of modified biomolecules such as proteins, DNA and RNA oligonucleotides containing desired properties useful for the creation of novel applications and nano-devices. During the last decades, chemical conjugation reactions have been applied in biology, molecular biology, and molecular medicine for various applications.
A "bioconjugate" refers to a molecular species that can be produced by living systems or synthetically by chemists or biologists. A bioconjugate is composed of at least two different molecular parts of biological origin. Bioconjugates used in organisms or cells are designed such that they are water soluble or that cells are able to compartmentalize them. For synthetic production of bioconjugates different bioconjugation chemistries are used.
Avidin-Biotin or Streptavidin-Biotin based techniques are widely used in biological and medical research. Flurescent conjugates of avidin and streptavidin can be used as amplification methods in flow cytometry, fluorescent imaging, in-sity hybridization, western blotting, ELISA and other microplate-based methods. Fluorescent conugates of both, avidin and streptavidin, are useful tools for the detection of biotinylated biomolecules.
Bioconjugate chemistry, often also called bioconjugation chemistry, is a research field that studies the linking of one molecule to another by chemical or biological means. Typically, the resulting complexes are formed from at least one biomolecule, however, several molecules may be conjugated together as well. In addition, purely synthetic conjugated molecules are possible as well.
The study and use of chemical conjugation reactions, now also known as bioconjugation reactions, has more recently evolved into an important research topic as can be monitored by the number of publications in this field. For example, site-specific bioconjugations of a multitude of biomolecules to proteins, DNA, RNA, and carbohydrates, or to each other, have been developed. The resulting conjugates are useful for applications such as ligand discovery, disease diagnosis, and high-throughput screening, in vivo imaging, sensing, catalysis, therapeutics, as well as cell targeting. More recently, polymer brushes linked with biotin moieties allowing for the development of streptavidin-mediated conjugation capture agents in NanoVelcro chips have been engineered. These conjugates are a new type of molecular probes for prenatal diagnostics (GEN July 2015).
General bioconjugation chemistry schemes are illustrated in figure 1. Typically, bioconjugation reactions are employed to couple biomolecules to surfaces or solid supports. Typical supports are beads, gold surfaces, nitrocellulose or dextran based arrays. The use of these chemistries allows the synthesis of oligonucleotide-, peptide-, or protein based libraries coupled to a solid support such as a micro-chip. Typical applications are screening or sequencing by hybridization. The conjugation of oligo-nucleotides to a support is shown as an example. However, peptides, carbohydrates and proteins can also be used in a similar fashion. Other nanostructures such as dendrimers, cyclodextrin or cellulose, modified or unmodified, are also often utilized as spacers or amplifying moieties, usually between the support and the biomolecule selected for conjugation.
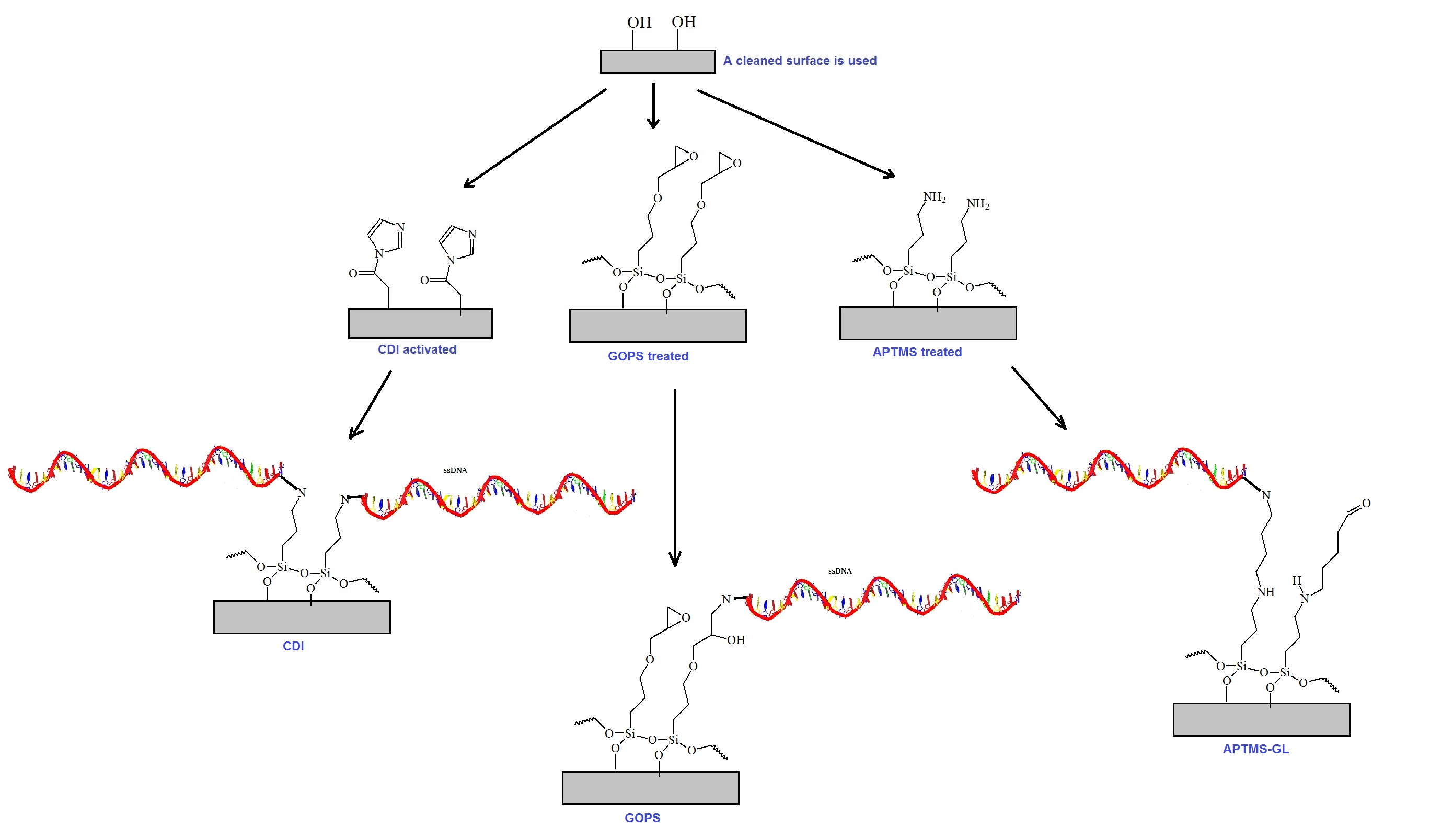
Figure 1: General schematic of bioconjugation chemistries. The conjugation of oligonucleotides to a solid support or surface is illustrated here. Three bioconjugation chemistries commonly used are reviewed. Carbonyl-diimidazole (CDI) activation to silanol groups is shown to the left. The surface functionalization technique using glycidooxypropyl-trimethoxy-silane (GOPS) is shown in the middle. And, the surface functionalization technique using 3-aminopropyltrimethoxysilane (APTMS) is shown at the right. Dendrimers or other branched polymers or other spacer molecules can also be used, often between the support and the molecule to be conjugated.
Bioengineering or Biological engineering is a new scientific field that applies engineering principles to biological systems. Broadly viewed, bioengineering can include elements of electrical, mechanical, computer science, materials, chemistry, biology and medical biology. Its main goal is to apply concepts and methods observed in biological systems to solve real-world problems in life sciences. Often medical biology is part of this endeavor. Bioengineering uses primarily knowledge gained from the fast developing field called “molecular biology.” A rapid development of many innovative techniques and methodologies pertinent to biological or medical applications using bioengineering principles leading to new applications in medicine, agriculture, and energy or electronic production occurred in recent decades. A new branch known as “biomimetics” strives to engineer new materials to mimic structures and functions of molecules found in living organisms using DNA, RNA, and protein molecules. The goal is the production or manufacture of new types of nano-materials, such as hydrogels,nano-particles, artificial proteins, antibodies, peptides, or dendrimers, and many others. However, the success of each conjugation reaction depends highly on its chemoselectivity under physiological conditions. Therefore, various chemoselective cross-linkers have been developed for the purposes of labeling and conjugation of selected molecules to functional groups available on target molecules. The following paragraphs describe several of these bioconjugation chemistries.
(1) Formation of amide bonds, including urea and thiourea moieties.
Typically amide bonds are formed through the reaction of an amino group with a carboxylic acid. Activator reagents are utilized to form the bonds more efficiently. Activator molecules such as 1-ethyl-3-(3-dimethylaminopropyl) carbodiimide hydrochloride (EDC), O-(benzotriazol-1-yl)-N,N,N',N'-tetramethyluronium hexafluorophosphate (HBTU), carbonyl-diimidazole (CDI) or similar compounds are commonly used as illustrated in figure 2A. Figures 2B and 2C show the reaction of the amino group with active esters. The reaction of amino groups with isocyanate or isothiocyanate produces a urea or thiourea backbone, respectively, as depicted in figure 1D. Many biomolecules including proteins have many amino and carboxyl groups at their molecular surface. Also, amino and carboxyl groups can be introduced to synthetic oligonucleotides and peptides with ease. Therefore, these chemistries are most widely used for the production of bioconjugates. Even some newly developed cross-linkers, for example, copper-free click chemistry based cross-linkers, use these types of reaction known to organic chemists for a long time, to modify biomolecules. Under standard conditions, these chemical reactions are normally quick and selective, forming stable amide bonds within a few hours. However, the majority of conjugation reactions are normally performed in neutral or slightly basic conditions, free of primary and secondary amines. EDC-mediated conjugations are an exception. In this case, reactions are best performed at slightly acidic conditions. Amide forming reactions are normally selective towards amino groups and are thermodynamically or kinetically controlled. However, isocyanates and isothiocyanates also react with the hydroxyl group to form carbamate and thiocarbamate bonds, respectively. Thus, these cross-linkers are also used to conjugate hydroxyl-containing molecules.
Figure 2. Amide formation reactions frequently used in bioconjugation.
(2) Formation of thioethers
The formation of thioesters is a widely used conjugation chemistry known for quite some time now. Reactions are very selective and specific since thiol groups are available in proteins that contain cysteine. Some proteins, such as members of the C3/α-2M thioester protein family, and peptides that contain lanthionine, contain natural interchain thioester bonds. The formation of thiols bonds in biomolecules is relative straight forward. Also, incorporation of thiol groups into synthetic oligonucleotides and peptides is done with the help of standard chemistries. An advantage of the thioester chemistry is that primary amines do not interfere with the reaction avoiding the formation of byproducts. Also, reactions can be performed at a broad pH range, ranging from pH 2 to 10. Therefore, many hetero-bifunctional cross-linkers are typically designed to contain one thiol-reactive group andoneamino-reactive group. Reactive compounds that form thioether bonds or linkages include thiol-maleimide (Figure 3A), and thiol-haloacetate (Figure 3B). The lesser electronegativity between the sulfur and hydrogen atoms, compared to the oxygen and hydrogen atoms, make the thiol group less polar than the hydroxyl group. The reductive dealkylation of thioethers generates thiols.
Figure 3. Frequently used bioconjugation thioether formation reactions.
(3) Conjugation reactions involving carbonyl group
The presence of carbonyl groups in biomolecules is very limited. However, these are important functional groups allowing the conjugation of saccharide moieties. Except for the aldehyde and ketone groups of the linear saccharides, hydroxyl groups at the anomeric position of the cyclic saccharides normally are not reactive enough and, therefore, are not available for bioconjugation reactions. In particular, 1,2-diol groups in saccharides can be specifically oxidized to aldehydes with the help of sodium periodate. Carbonyl groups can be introduced to biomolecules using cross-linkers such as N-succinimidyl-4-formylbenzamide (S-4FB). Carbonyl groups are reactive towards primary amines, including hydrazine and oxyamine groups, whereas thiol and hydroxyl groups usually don’t interfere with the reaction. The reaction of a carboxyl group with a primary amine, hydrazine or oxyamine form Schiff bases, hydrazone or oxime moieties, respectively, as illustrated in figure 4. Oxime groups are quite stable and exist as two stereoisomers. However, Schiff bases and hydrazones are more labile, especially in acidic solutions or conditions. Therefore, the Schiff base is typically reduced to a secondary amine with the help of sodium cyanoborohydride, called reductive amination, as depicted in figure 4A. Furthermore, hydrazones can also be reduced and are typically stabilized by adding an aromatic group or groups to the resulting compounds in proximity to the hydrazone group.
Figure 4. Conjugation reactions involving carbonyl groups.
(4) Thiol-exchange reactions
Thiol-exchange reactions are widely used because these reactions produce cleavable conjugates. Disulfide bonds are created between the conjugated molecules. Disulfide bonds containing conjugates can be cleaved via reduction by thiol reductases in tissue or cells. Furthermore, disulfide bonds can also be chemically cleaved in vitro using reducing agents such as mercaptoethanol (2-ME), tris(2-carboethyl)phosphine (TCEP), or dithiothreitol (DTT). However, the reactions for the formation of conjugates are usually slow but very selective towards the thiolated molecules. Amino or hydroxyl groups don’t usually interfere to produce byproducts. Although sometimes methanethiosulfonate reagents are used, 2-pyridyldithiol reagents are the most widely used thio-exchange cross-linkers, illustrated in figure 5.
Figure 5. The thiol-exchange reaction of 2-pyridyldithiol based cross-linkers.
(5) Click chemistry and tetrazine ligation
Click chemistry has gained a huge momentum in bioconjugation due the development of the reagents recently. Traditionally, click chemistry refers to the reaction of an alkynyl compound with an azido compound by the catalysis of Cu(I) (Figure 6A). Although this reaction is very selective and clean, coordination of Cu(I) with many ligands makes the complete removal from the conjugate problematic. Furthermore, the toxicity of copper limits the application of click chemistry in many conjugation chemistries. Copper-free click chemistry overcomes this problem. Here, cyclooctyne is used instead of common alkynes, the tension in the 8-member ring promotes the click reaction, no longer requiring Cu(I) catalysis. Also, since recently needed reagents became available at a lower price, the copper-free click chemistry became very popular in recent years. Frequently-used cyclooctynyl reagents include the cross-linkers dibenzocyclooctynyl (DBCO) and bicyclo[6.1.0]nonynyl (BCN). In addition, the Diels-Alder reaction and reverse Diels-Alder reaction has been studied for years as a conjugation chemistry. A recent successful development is the conjugation reaction of trans-cyclooctene to tetrazines, called, a tetrazine ligation (Figure 6B). Here, the tension in the trans-cyclooctene ring allows the reaction to occur at room temperature, relatively fast and without the need for a catalyst.
Figure 6. Click chemistry (A) and tetrazine ligation (B)
(6) Photoreactive cross-linkers
Photoreactions are only frequently performed in bioconjugation reactions. However, they are occasionally used for in-vivo or in-vitro crosslinking reactions. Photoreactive cross-linkers commonly used are azidophenyl or diazirine compounds. Upon exposure to ultraviolet light, these compounds are activated and react with electron-donating groups, most frequently amino group, to produce conjugates (Figure 5). The reactions are normally not very fast and unselective, and may have multiple pathways. Also, products are not very clean. One advantage of photocrosslinking is that biomolecules that are selected to be conjugated can be mixed prior to the conjugation reaction, and the start of the reaction can be controlled.
Figure 7. Photoreactions frequently used in bioconjugation.
References
Advances in Bioconjugation: http://www.ncbi.nlm.nih.gov/pmc/articles/PMC2901115/
BC Bioconjugate Chemistry Journal: http://pubs.acs.org/journal/bcches
Chemical modifications and bioconjugate reactions of nanomaterials for sensing, imaging, drug delivery and therapy