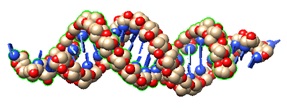
In the endogenous regulatory process called RNA interference (RNAi), short double-stranded RNA oligonucleotides cause sequence-specific posttranscriptional silencing of genes. Small interference RNA (siRNA) has now emerged as a promising therapeutic strategy for the development of novel drugs. However, for successful systemic delivery of siRNA therapeutics, siRNAs must penetrate cellular barriers to reach their targets in the cytoplasm.
The commonly utilized intravenous injection of siRNA poses a significant challenge. After intravenous injection, the blood circulation distributes siRNAs to organs where, at the same time, the siRNA undergoes elimination. When arriving in an organ, siRNA enters the interstitium by leaving the intravascular space within a blood vessel. From the tissue interstitium, siRNA is transported across the interstitial space to target cells. The target cell internalizes siRNA via endocytosis. This process encapsulates siRNA in endocytic vesicles that fuse with endosomes. After entering the cell, siRNA must escape from the endosomes into the cytosol for RISC loading.
Among other approaches, cell-penetrating peptides enable systemic siRNA delivery. Utilizing a cell-penetrating peptide recently, Tönges et al. developed a more straightforward method to deliver siRNA into rat neurons.
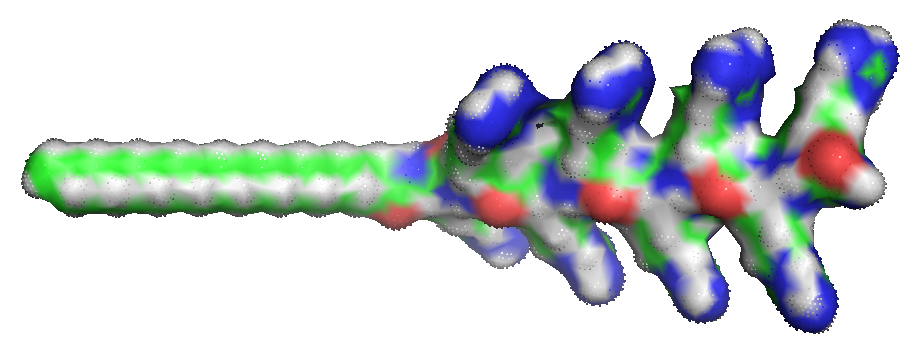
A cell-penetrating peptide modified with a stearyl moiety known as stearyl-R8 enables peptide-mediated transfection of cells and possible organelles.
| 
| 
|
| Stearly-R8 (2) | siRNA (1) |
Tönges et al. 2006 showed that a stearylated octaarginine peptide (stearyl-R8) and artificial virus-like particles facilitate the transfection of siRNA into primary rat neuron cells.
The arginine peptide-siRNA complex shuttles the oligonucleotide through the cell membrane into the cell. Complexing siRNA oligonucleotides with stearyl-R8 removes the need for the covalent attachment of the siRNA to the arginine carrier peptide.
Tönges et al. found that peptide-mediated transfection with stearyl-R8 and the polymer/lipid-based cellular delivery with artificial virus-like particles are both easy to handle. Only pipet-based mixing of siRNA with the selected transfection reagent is needed. No further chemical modifications of the siRNA are required.
For an efficient transfection experiment, the research group used synthetic Stearyl-R8 to prepare the Stearyl-R8:siRNA mixture with a cation-to-anion charge ratio 2:1.
This approach may also enable specific targeting of organelles, for example, mitochondria, lysosomes, the endoplasmic reticulum (ER), and the Golgi apparatus. All these organelles are enclosed with membranes or have specialized membrane-engulfed compartments embedded within the cytoplasm. Since they all perform specific roles in the cell, their dysfunction can lead to various pathophysiologies.
Reference
Geng J, Wang J, Wang H. Emerging Landscape of Cell-Penetrating Peptide-Mediated Organelle Restoration and Replacement. ACS Pharmacol Transl Sci. 2023 Jan 16;6(2):229-244. [PMC]
Tönges, L, Lingor, P, Egle, R, Dietz, GP, Fahr, A and Bahr, M (2006). Stearylated octaarginine and artificial virus-like particles for transfection of siRNA into primary rat neurons. RNA 12: 1431-1438. [PMC]
Wang J, Lu Z, Wientjes MG, Au JL. Delivery of siRNA therapeutics: barriers and carriers. AAPS J. 2010 Dec;12(4):492-503. [PMC]
https://www.biosyn.com/tew/MITO-Porters-Enable-Delivery-of-Antisense-Drugs-to-Mitochondria.aspx
https://www.biosyn.com/tew/Cell-penetrating-peptides-for-the-delivery-of-siRNA-into-cells.aspx
https://www.biosyn.com/tew/cell-penetrating-or-trojan-peptides-cpps.aspx
https://www.biosyn.com/tew/cell-penetrating-peptides.aspx
---...---
" Bio-Synthesis provides a full spectrum of high quality custom oligonucleotide modification services including 5'-triphosphate and back-bone modifications, conjugation to fatty acids, biotinylation by direct solid-phase chemical synthesis or enzyme-assisted approaches to obtain artificially modified oligonucleotides, such as BNA antisense oligonucleotides, mRNAs or siRNAs, containing a natural or modified backbone, as well as base, sugar and internucleotide linkages.
Bio-Synthesis also provides biotinylated mRNA and long circular oligonucleotides".
---...---