Labeling and Protecting Groups used in Peptide Synthesis
In a peptide, each monomer unit in the sequence chain is known as an amino acid residue. The term residue refers to the fact that each amino acid in a peptide or protein sequence has lost one molecule of water during polymerization or synthesis. In peptides and proteins, the number of water molecules lost is one less than the number of residues. The peptide Ala-Val-Met, or AVM, or more precise L-alanyl-L-valyl-L-methionine has the following structure:
Figure 1: Structure and model of AVM. The model of the structure is displayed as a stick model with dots showing the Van Der Waals spheres.
Solid phase peptide synthesis is now commonly used for the formation of peptide bonds. To successfully link amino acids together to form a multitude of peptides protection strategies are needed. For peptide synthesis, protecting strategies have been developed utilizing a variety of temporary protecting groups that can be selectively removed either during or after synthesis. The Fmoc (9-fluorenyl-methoxycarbonyl)-group has become the most widely used N-terminal protection group in Fmoc-peptide synthesis strategies.
Figure 2: Structure and model of Fmoc-Cl.
Figure 3: Structural models of Fmoc-Amino Acid and Fmoc-Dipeptide.
Since amino acid side chains represent a broad range of organic functional groups, a collection of different specific protecting groups has been developed over the years. Most of these are now commercially available. The following tables contain a list of protecting groups.
Delta Mass and Masses of some
Protecting Groups and Adducts
|
Δm and m
|
Group
|
|
14.027
|
Methylation, Me: Adding CH2
|
|
15
|
Methyl, CH3
|
|
22
|
Sodium, Na
|
|
28.104
|
Formyl, CHO
|
|
28.054, 29
|
Ethyl, CH3CH2
|
|
38
|
Potassium, K
|
|
42.037
|
Acetyl, Ac
|
|
56.108, 57.1
|
tBu; tert-Butyl, C4H9
|
|
90.126
|
Anisyl
|
|
90.1, 91.1
|
Bzl; Benzyl, C7H7
|
|
96.009
|
TFA, trifluoroacetyl
|
|
100.117
|
Boc, t-Butyloxycarbonyl
|
|
104.108
|
Bz, Benzoyl
|
|
106.191
|
Thioanizyl, thiocresyl
|
|
222.243
|
Fmoc
|
|
243.3
|
Trt, Trityl; C17H15
|
|
252.3. 253.3
|
Pbf
|
|
266.361, 267.4
|
Pmc
|
See also ABRF Delta Mass: https://abrf.org/delta-mass
Protecting groups most often used in organic synthesis and solid phase peptide synthesis (SPPS)
|
Protecting Group
|
Groups protected
|
Introduction
|
Cleavage
|
|
tert-Butyl
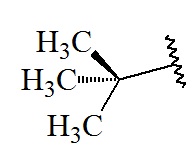
C4H9; RW: 57.1
|
-CO2H
-OH
-SH
|
or
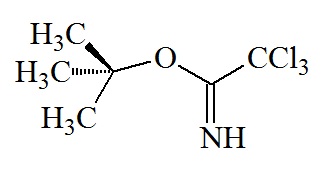
|
Moderately strong acid (e.g.
95% CF3CO2H
or
HCl in dioxane)
|
|
Benzyl (Bzl)
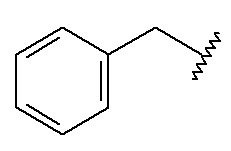
C7H7; RW: 91.1
|
-CO2H
-OH
-SH
|
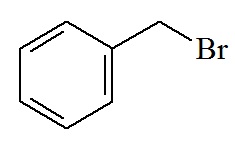
|
Hydrogenolysis
(e.g. H2 / Pd)
or Strong acid
(e.g. HF or
HBr in CF3CO2H)
(or for SH protection Na/NH3)
|
|
Trityl (Trt)
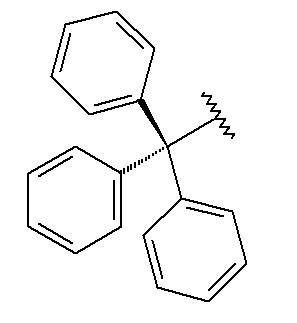
C17H15; RW: 243.3
|
-CONH2
-SH
(His imidazole
 ) )
(-NH2)
|
|
Mild acid
(e.g. CH3CO2H
or
5% CF3CO2H in organic solvent)
|
|
Methyl, ethyl
CH3, CH3CH3;
RW: 15; 29
|
- CO2H
|
CH3OH or C2H5OH with acid catalyst (HCl or
|
Basic saponification
(e.g.
CH3OH/NaOH)
|
|
2,2,5,7,8-pentamethyl-chroman-6-sulphonyl (Pmc)
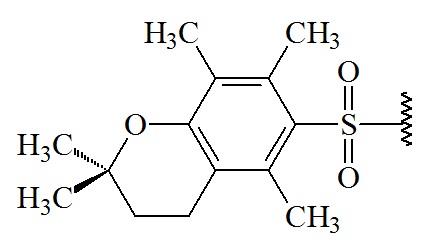
C14H19O3S; RW: 267.4
|
-NHC(NH2)=NH
(Arg)
|
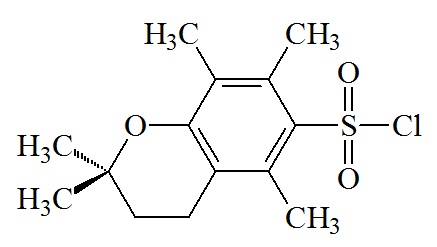
|
Moderately strong acid
(e.g. CF3CO2H)
|
|
2,2,4,6,7-pentamethyl-dihydro-benzofuran-5-sulfonyl (Pbf)
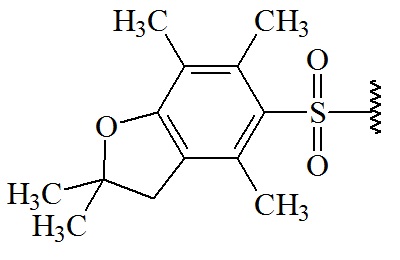
C13H17O3S; RW: 253.3
|
-NHC(NH2)=NH
(Arg)
|
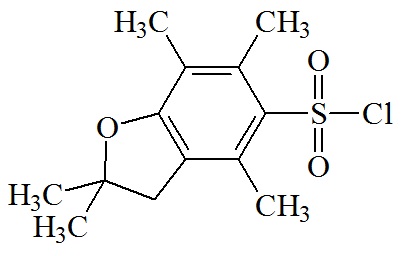
|
Moderately strong acid
(e.g. CF3CO2H)
|
-.-