The 2,2,7-trimethylguanosine (TMG or m3G) cap is one of the earliest identified RNA modifications. The TMG-cap ensures nuclear localization of small nuclear RNAs (snRNAs).
Research during recent decades has provided a better understanding of the biological roles of base and ribose modifications, as well as cellular degradation pathways. Advances in enzymatic synthesis, RNA modifications, and cellular delivery vehicles enabled the administration of exogenous mRNA into cells to express proteins in situ. A few examples illustrating this progress are stem cell reprogramming, vaccination, and the expression of therapeutic proteins.
The capping machinery of RNAs, particularly mRNAs, is quite complicated. Cap structures of the type m7GpppNmpNm are at the 5’-ends of nearly all eukaryotic cellular and viral mRNAs. The m7GpppNmpNm recruits cellular proteins and mediates cap-related biological functions such as pre-mRNA processing, nuclear export, and cap-dependent protein synthesis. Also, the 2’-O-methylated cap is an identifier of self RNA in the innate immune system against foreign RNA. The discovery of the cytoplasmic capping machinery suggested an additional control network. For example, Singh et al., in 1989, characterized the cap structure of human U6 snRNA and showed that the gamma phosphate of the 5' guanosine triphosphate is methylated.
Structures of Uncapped and Capped RNA including the TMG Cap
| 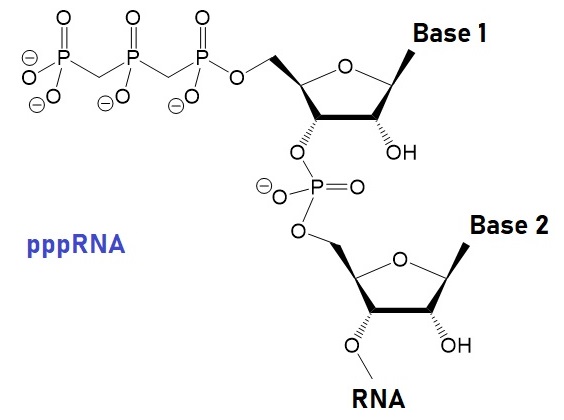
Uncapped RNA |  γ-methyl snRNA cap |
|  TMG snRNA CAP, m3GpppRNA |

m7G mRNA caps, m7GpppRNA
|
The structural models of a TMG-capped dinucleotide bound to the nuclear import receptor snurportin 1 domain are illustrated below. Snurportin 1 interacts with the m3G-cap enhancing the m3G-cap-dependent nuclear import of U snRNPs in Xenapus laevis oocytes and digitonin-permeable HeLa (human) cells.
| | 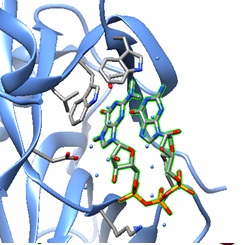 | 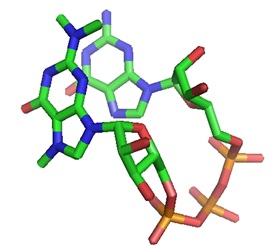 |
| A: Ribbon plot of the crystal structure of the m3G-Cap-binding domain of snurportin1 in complex with a m3GpppG-Cap dinucleotide (PDB ID 1X5K). The m3GpppG-Cap dinucleotide is shown in surface mode ( Strasser et al. [ PMC ]). | B: The m3GpppG-Cap dinucleotide is shown in its binding pocket. | C: The m3GpppG dinucleotide is depicted in stick mode with nitrogen atoms in blue, carbons in grey, oxygens in red and phosphorus atoms in yellow. |
Do TMG-capped oligonucleotides accumulate in the nucleus?
Yes, according to a recent paper by Moreno and others.
RNA oligonucleotides capped with the 2,2,7-trimethylguanosine (TMG or m3G) cap can direct nuclear accumulation of a cargo protein in mammalian cells after cytosolic delivery using a transfection reagent. Moreno et al., in 2009, reported the use of the m3G- or TMG-cap as an adaptor molecule that, when attached to oligonucleotides, enables targeted transport of large-size cargo molecules into the nucleus. Furthermore, TMG-capped 2′-O-methyl RNA antisense oligonucleotides (ASOs) showed increased efficiency in a splice correction assay. Moreno et al. suggested that synthetically capped oligonucleotides may selectively interfere with splicing events, potentially resulting in therapeutic clinical applications (Moreno et al.).
A TMG-cap structure enables nuclear import!
The spliceosomal small nuclear ribonucleoproteins snRNPs U1, U2, U4, and U5 depend on a complex nuclear localization signal (NLS) for their nuclear import. The NLS comprises the 5'-2,2,7-terminal trimethylguanosine (m3G or TMG) cap structure of the U snRNA and the Sm core domain. When injected into Xenopus laevis oocyte nuclei, U snRNA precursor molecules temporarily concentrate in Cajal bodies before their export (Suzuki et al.)
The TMG-cap structure is essential for telomere maintenance!
The TMG-cap plays a central role in recruiting telomerase to telomeres and engaging Cajal bodies in telomere maintenance. Many RNA molecules contain the TMG-cap at the 5’-end.
Buemi et al. recently showed that the formation of the TMG cap structure at the human telomerase RNA 5′-end by the Trimethylguanosine Synthase 1 (TGS1) is needed for recruiting telomerase to telomeres and engaging Cajal bodies in telomere maintenance. Depletion of the enzyme TGS1 with sinefungin blocked telomerase recruitment to telomeres. The result was exonuclease 1 (EXO1)-mediated formation of single-stranded 3′-telomere overhangs that engage in RAD51-dependent homologous recombination and APB formation and the activation of key features of the ALT pathway in telomerase-positive H1299 cells and lung tumor organoids.
The alternative lengthening telomere pathway (ALT pathway) refers to the telomerase-independent, recombination-dependent process that extends telomeres. APB refers to the ALT-associated promyelocytic leukemia nuclear body. Recent findings indicate a critical role for 2,2,7-TMG capping in telomerase-dependent telomere maintenance to restrict the formation of telomeric substrates conducive to ALT. This pathway is relevant in telomere-related diseases such as cancer and aging.
The Nuclear Import Machinery
Transporting biomolecules from the cytoplasm to the nucleus is critical for cell physiology and pathology. The nuclear envelope is a significant barrier to delivering therapeutic molecules into the nucleus. The large size of nucleic acids makes delivering therapeutic molecules into the nucleus difficult for medical applications. The deregulation of the import and export machinery is a cause of many diseases.
The nuclear envelope comprises control gates known as nuclear pore complexes (NPCs) that selectively regulate nuclear transport. Small molecules like metabolites and ions can diffuse through the NPC. However, molecules bigger than 40 kDa, like most proteins, RNA, ribosomal subunits, and viral particles, require an active transport mechanism. Soluble carrier molecules belonging to the karyopherin-β family, including importins, exportins, and transportins, are part of the transporting machinery.
The NPC is a large protein complex fusing the internal and external nuclear membrane forming an aqueous channel. Approximately 2000 to 5000 NPCs are present in a vertebrate cell. Extensive studies of NPC by electron microscopy and proteomic approaches are available now (Cautain et al., 2014).
The Ran system provides the energy source for efficient cargo transport. In particular, the Ran-GTP/GDP gradient across NPC regulates the transportation of cargo molecules between the nucleoplasm and the cytoplasm. RanGTP binds to a carrier importin and helps to release the cargo in the nucleus. After transporting cargo to the cytoplasm, the exportin-RanGTP-cargo complex is hydrolyzed, releasing the cargo molecule.
Large cargo molecules need a nuclear localization signal (NLS) to access the nucleus. Classical import signals (cNLS) contain a short stretch of basic residues. The adaptor protein importin-α recognizes a cargo molecule containing the cNLS. For interaction with NPS, the complex binds to importin-β. RanGTP mediates the translocation of the cargo into the nucleus. However, non-classical NLSs do not need an adaptor protein by binding directly to a transporter protein. Table 1 lists examples of different nuclear transport signals.
Table 1: Examples of nuclear transport signals.
| Amino acid sequence | Protein | Signal |
| PKKKRKV | SV40 T antigen | Classical NLS |
| KRx{10}KKKL | Nucleoplasmin | Bipartite NLS |
| VRILESWFAKNIENPYLDT | Matα2 | Polar/nonpolar residues NLS |
| PAAKRVKLD | c‐Myc | cMyc‐NLS |
| YNDFGNYNNSSNFGPMKGGNFGGRSSGPYGGGGQY | hnRNP A1 (M9 sequence hydrophobic subclasses) | hPY‐NLS |
| KVSRRG‐GHQNSYKPY | hnRNP D (basic enriched) | bPY‐NLS |
| RQARRNRRRRWR | VIH Rev protein | Arginine‐rich NLS |
| DNSQRFTQRGGGAVGKNRRGGRGGNRGGRNNNSTRFNPLAK | Nab2p | Arginine/glycine‐rich NLS |
| KTPGKKKKGK | Parathyroid hormone‐related protein (PTHrP) | Lysine‐rich‐NLS |
| QDLNSTAAPHPRLSQYKSKYSSLEQSERRRRL | Snurportin1 | UsnRNPs‐NLS |
| LPPLERLTL | HIV Rev | Hydrophobic‐NES |
| LALKLAGLKI | PKI | NES |
| LCQAFSDVIL | Cyclin B1 | NES |
| LQKKLEELEL | MAPKK | NES |
| LAEMLEDLHI | NMD3 | NES |
( Source: Cautain et al., made freely available through PubMed Central as part of the COVID-19 public health emergency response. It can be used for unrestricted research re-use and analysis in any form or by any means with acknowledgement of the original source, for the duration of the public health emergency. )
The non-covalent or covalent complexation of an NLS peptide with a plasmid DNA or an oligonucleotide promotes nuclear delivery. Also, transfection is possible by combining plasmid DNA with polyethylimines (PEIs) conjugated to an NLS peptide.
Small nuclear RNAs (snRNAs) as part of small nuclear RNA binding proteins (snRNPs) are mainly found in U1, U2, U5, and U4/U6. snRNAs are transcribed by RNA polymerase II, and co-transcriptionally capped with an m7G-Cap. Uridine-rich, small nuclear ribonucleoproteins (U snRNPs) involved in pre-mRNA splicing contain the TMG-cap as an NLS signal.
The heterodimeric cap-binding complex (CBC) recognizes the m7G-Cap. It recruits the phosphorylated adaptor of export protein (PHAX), further recognized by an export receptor CRM1 for cytoplasmic localization and maturation. Next, a complex of proteins assembles around the SM core domain of snRNAs, creating a platform for trimethylguanosine synthase 1 (TGS 1). TGS 1 catalyzes the methylation of the m7G-cap to the m3G-cap (TMG-cap). The TMG-cap is recognized by the adaptor protein surportin 1. During the final maturation of snRNPs, this complex binds to importin-β for nuclear relocalization.
Sm-proteins received their name from human autoimmune serum initially used in their identification. In human cells, the assembly of the snRNAs with the Sm-proteins is highly specific and mediated by a large protein complex that includes the SMN protein (survival of motor neurons).
Reference
Buemi, V., Schillaci, O., Santorsola, M. et al. TGS1 mediates 2,2,7-trimethyl guanosine capping of the human telomerase RNA to direct telomerase dependent telomere maintenance. Nat Commun 13, 2302 (2022). [nature]
Cautain B, Hill R, de Pedro N, Link W. Components and regulation of nuclear transport processes. FEBS J. 2015;282(3):445-462. [PMC]
Furuichi Y. Discovery of m(7)G-cap in eukaryotic mRNAs. Proc Jpn Acad Ser B Phys Biol Sci. 2015;91(8):394-409. [PMC]
Honcharenko, M., Bestas, B., Jezowska, M., Wojtczak, B. A., Moreno, P. M. D., Romanowska, J., Baechle, S. M., Darzynkiewicz, E., Jemielity, J., Smith, C. I. E., and Stroemberg, R. (2016) Synthetic m3G-CAP attachment necessitates a minimum trinucleotide constituent to be recognised as a nuclear import signal. RSC Adv. 6, 51367-51373. [RSC]
Pedro M. D. Moreno, Malgorzata Wenska, Karin E. Lundin, Örjan Wrange, Roger Strömberg, C. I. Edvard Smith, Nucleic Acids Research, Volume 37, Issue 6, 1 April 2009, Pages 1925–1935. [NAR]
Moteki S, Price D. Functional coupling of capping and transcription of mRNA. Mol Cell. 2002 Sep;10(3):599-609. [Molecular-Cell]
Ramanathan A, Robb GB, Chan SH. mRNA capping: biological functions and applications. Nucleic Acids Res. 2016;44(16):7511-7526. [PMC]
Shatkin AJ. Capping of eucaryotic mRNAs. Cell. 1976 Dec;9(4 PT 2):645-53. [Pubmed]
Shatkin A.J., Manley J.L., The ends of the affair: capping and polyadenylation. Nat. Struct. Biol. 2000, 7, 838-842. [Pubmed]
Shih DS, Dasgupta R, Kaesberg P. 7-Methyl-guanosine and efficiency of RNA translation. J Virol. 1976 Aug;19(2):637-42. doi: [PMC]
Singh R, Reddy R. Gamma-monomethyl phosphate: a cap structure in spliceosomal U6 small nuclear RNA. Proc Natl Acad Sci U S A. 1989 Nov;86(21):8280-3. [PMC]
A, Dickmanns A, Lührmann R, Ficner R. Structural basis for m3G-cap-mediated nuclear import of spliceosomal UsnRNPs by snurportin1. EMBO J. 2005;24(13):2235-2243. [ PMC ].
Suzuki T, Izumi H, Ohno M. Cajal body surveillance of U snRNA export complex assembly. J Cell Biol. 2010;190(4):603-612. [JCB]
Zhang JM, Yadav T, Ouyang J, Lan L, Zou L. Alternative Lengthening of Telomeres through Two Distinct Break-Induced Replication Pathways. Cell Rep. 2019 Jan 22;26(4):955-968.e3. [PMC]
---...---
Bio-Synthesis provides a full spectrum of oligonucleotide and peptide synthesis including bio-conjugation services as well as high quality custom oligonucleotide modification services, back-bone modifications, conjugation to fatty acids and lipids, cholesterol, tocopherol, peptides as well as biotinylation by direct solid-phase chemical synthesis or enzyme-assisted approaches to obtain artificially modified oligonucleotides, such as BNA antisense oligonucleotides, mRNAs or siRNAs, containing a natural or modified backbone, as well as base, sugar and internucleotide linkages.
Bio-Synthesis also provides biotinylated mRNA and long circular oligonucleotides.
---...---