Heart disease continues to cause a high rate of mortality in the U. S. as it accounts for ~20% of all deaths. Despite investing heavily on modifying diet, ~697,000 individuals may succumb to the disease annually. One of the major contributing factors is the accumulation of plaques within the coronary arteries (atherosclerosis), reducing the supply of oxygen to cardiac muscles. Oxygen plays a vital role in converting energy stored in nutrients (ex. glucose) into ATPs, the main currency used by cells to enable energy-requiring tasks. An insufficient level of oxygen may cause cardiac muscles to underperform, potentially leading to stroke, artery disease, myocardial infarction (heart attack), etc.
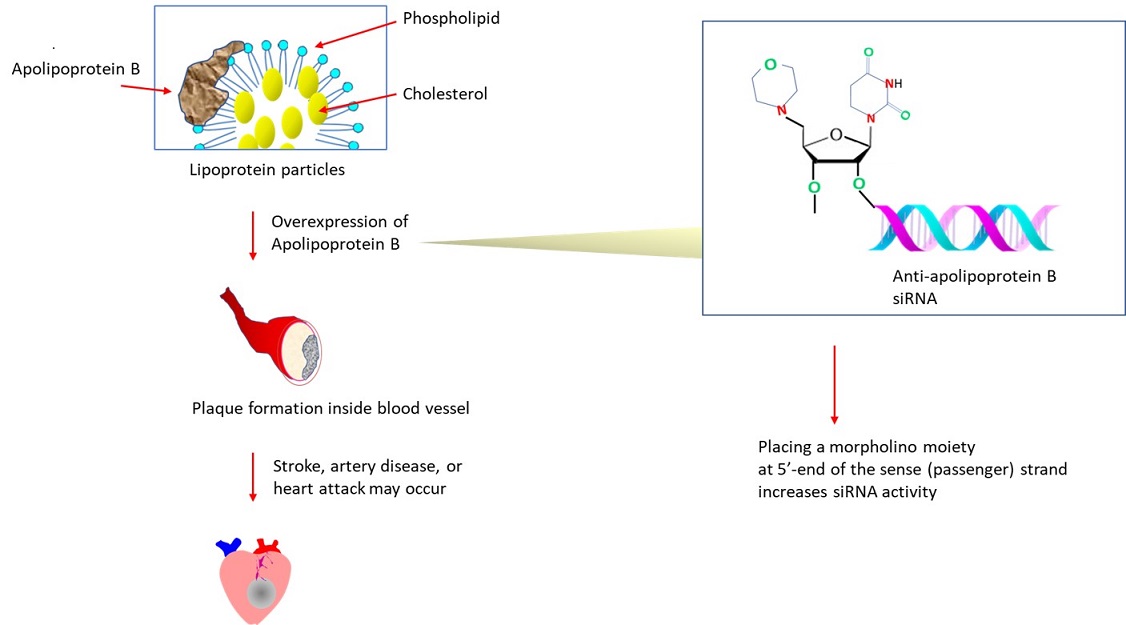
The plaques found inside blood vessels are comprised of lipid molecules, fibrous tissues, lipid-containing cells (macrophage-like), etc. As lipids (ex. cholesterol, triglycerides) are insoluble, they (~3000 to 6,000 molecules) are carried in spherically shaped ‘lipoprotein particles’ in plasma. The outer shell of the transport particle consists of apolipoprotein B, phospholipid, etc. Apolipoprotein B also serves as the ligand for the receptor that internalizes low density lipoprotein (LDL) particles via clathrin-mediated endocytosis. Within the endosome, low pH triggers a conformational change of the receptor, releasing LDL to recycle. Alternatively, it may get degraded in the lysosome after binding to PSCK9, which blocks its conformational change. A key determinant for plaque formation is the overexpressed apolipoprotein B. An altered expression (reduced) of apolipoprotein B may also play a role in liver cancer by upregulating metastasis-causing genes or downregulating tumor suppressor genes (Lee et al., 2018).
The last decade has seen a marked rise in the number of siRNA-based oligonucleotide therapeutics approved by the FDA (U. S. Food & Drug Administration). RNA interference refers to the post-transcriptional mechanism of silencing gene expression by cleaving mRNA for degradation. Originally discovered in nematode as well as in plants, the mechanism of RNA interference has subsequently been exploited for translational medicine. Critical to this endeavor is the ability of chemically synthesized siRNAs to elicit RNA interference in vivo (Elbashir et al., 2001). As the siRNA therapeutics rely on Watson-Crick base pairing for target recognition, it is highly specific.
Nonetheless, several aspects of the siRNA therapeutics may need further improvement. One area concerns its delivery as siRNAs are highly charged and bulky, making it difficult to translocate across the cell membrane. To facilitate, various delivery vectors are being explored, which include peptide, carbohydrate, lipid, and nanoparticle (Roberts et al. 2020).
Another potential issue concerns side effects caused by off-targeting. To scan the mRNA (to find complementary sequence), short siRNA duplex generated by the ribonuclease Dicer are strand-separated and loaded to RNA-induced silencing complex (RISC). In principle, as the siRNA duplex consists of two complementary strands, either the guide (antisense) strand or passenger(sense) strand could be loaded. The loading of the passenger strand may lead to the cleavage of an unintended mRNA.
Previous works suggest that the interaction of 5’-phosphate of the antisense (guide) strand and ‘MID domain’ of Argonaute 2 (of RISC complex) may facilitate strand selection (Elkayam et al., 2012). It inspired the testing of various modifications at the 5’ end of sense (passenger) strand (ex. 5’-O-methylation, 5’-unlocked nucleic acid unphosphorylated monomer) to ‘suppress’ its loading (Chen et al., 2007; Snead et al., 2013).
In another strategy, the effect of blocking the 5’ phosphorylation of the sense strand with morpholino was investigated (Kumar et al., 2013). The researchers at Alnylam Pharmaceuticals (United States) have developed siRNA targeting apolipoprotein B, which contains a morpholino group at the 5’ end of the sense or antisense strand. Reaction steps to chemically synthesize 5’-morpholino 2′-OMe phosphoramidites are described in detail in the report (Kumar et al., 2013). Placing morpholino on the sense strand improved the loading of anti-apolipoprotein B siRNA to the RISC complex, increasing the RNA interference activity.
The key to preventing epidemic is the ability to diagnose the infected early to preempt further propagation. For this, Bio-Synthesis, Inc. provides primers and probes (as well as synthetic RNA control) for COVID-19 diagnosis via RT-PCR assay. It specializes in oligonucleotide modification and provides an extensive array of chemically modified nucleoside analogues (over ~200; ex. morpholino) including bridged nucleic acid (BNA) in addition to mRNA synthesis. A number of options are available to label oligonucleotides (DNA or RNA) with fluorophores either terminally or internally as well as to conjugate to peptides or antibodies. It recently acquired a license from BNA Inc. of Osaka, Japan, for the manufacturing and distribution of BNANC, a third generation of BNA oligonucleotides. To meet the demands of therapeutic application, its oligonucleotide products are approaching GMP grade. Bio-Synthesis, Inc. has recently entered into collaborative agreement with Bind Therapeutics, Inc. to synthesize miR-21 blocker using BNA for triple negative breast cancer. The BNA technology provides superior, unequalled advantages in base stacking, binding affinity, aqueous solubility and nuclease resistance. It also improves the formation of duplexes and triplexes by reducing the repulsion between the negatively charged phosphates of the oligonucleotide backbone. Its single-mismatch discriminating power is especially useful for diagnosis (ex. FISH using DNA probe). For clinical application, BNA oligonucleotide exhibits lesser toxicity than other modified nucleotides.
https://www.biosyn.com/tew/Of-Target-Effects-in-small-interfering-RNA-or-siRNA.aspx
https://www.biosyn.com/tew/Thiophosphoramidate-Morpholinos,-A-New-Class-of-Antisense-Oligonucleotides.aspx
https://www.biosyn.com/tew/Peptide-conjugated-Antisense-Oligonucleotides-for-Exon-Skipping-Therapeutics.aspx
https://www.biosyn.com/tew/Phosphoryl-guanidine-oligo-2%E2%80%B2-O-methylribonucleotides-in-neutral-therapeutic-oligonucleotides-enable-enhanced-cell-penetration!.aspx
References
Elbashir SM, Tuschl T, et al.; Duplexes of 21-nucleotide RNAs mediate RNA interference in cultured mammalian cells. Nature. 411:494-8 (2001). PMID: 11373684 [PubMed]
Elkayam E, Joshua-Tor L, et al.; The structure of human argonaute-2 in complex with miR-20a. Cell. 150:100-10 (2012). PMID: 22682761 [PMC]
Kumar P, Manoharan M, et al.; 5'-Morpholino modification of the sense strand of an siRNA makes it a more effective passenger. Chem Commun (Camb). 55:5139-5142 (2019). PMID: 30977478 [ChemComm]
Lee G, Yim SY, et al.; Clinical significance of APOB inactivation in hepatocellular carcinoma. Exp Mol Med. 50:1-12 (2018). PMID: 30429453 [Nature]
Roberts TC, Langer R, et al.; Advances in oligonucleotide drug delivery. Nat Rev Drug Discov. 19:673-694 (2020). PMID: 32782413 [PMC]
Snead NM, McCaffrey AP, et al.; 5'-Unlocked Nucleic Acid Modification Improves siRNA Targeting. Mol Ther Nucleic Acids. 2:e103 (2013). PMID: 23820891 [PMC]