New drugs, such as oligonucleotide-based antisense drugs, peptide-drug conjugates, or lipid-drug conjugates, can potentially treat or cure many diseases. However, the drugs must reach their specific target and have improved pharmacokinetics to function as desired. The conjugation of drugs to macromolecular carriers improves the pharmacokinetics of the drug. However, stable conjugates can have several limitations:
(1) loss of the potency of the drug,
(2) confining the drug to the extracellular space, and
(3) requirement for circulating conjugates.
To improve the pharmacokinetics of drug conjugates, Santi et al. described conjugation linkers with highly tunable cleavage rates. The research group reviewed a set of tunable release linkers with half-lives ranging from hours to up to one year. Santi et al. suggest that these releasable linkers provide several benefits, including lower Cmax, the highest concentration of a drug in the blood, cerebrospinal fluid, or target organ after a dose is given, and the pharmacokinetic coordination of drug combinations. The multiple developed linkers have cleavage rates predetermined by the acidity of a C-H bond on the linker. A hydroxide-catalyzed β-elimination reaction releases the native, active drug.
Electron-withdrawing groups attached to the ionizable C-H group control the acidity of the linker. The linkers described by Santi et al. enable the construction of drug conjugates with predictable, tunable release kinetics over long periods useful in vitro and in vivo environments. Figure 1 shows a chemically releasable carbamate linker.
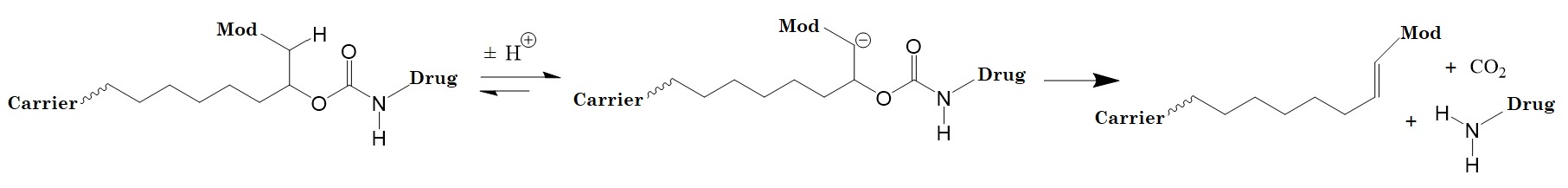
Figure 1: A chemically releasable carbamate linker. A macromolecular carrier is attached to a linker which is attached to the drug or prodrug via a carbamate group. The β-carbon has an acidic C-H group and contains an electron-withdrawing “modulator” that controls the pKa of that C-H group. Removing a proton initiates rapid β-elimination, cleaving the C-O of the linker–carbamate bond. A further loss of CO2 provides the free drug or prodrug and a substituted alkene. The drug release rate is proportional to the acidity (pKa) of the proton adjacent to the modulator. The chemical nature of the modulator controls the pKa and the release of the drug.
As a next step, the research group tested a carbamate containing Ne-2,4-dinitrophenyl-l-lysine [Lys(DNP)OH] and various electron-withdrawing pKa modulators. Figure 2 illustrates the releasing reaction.
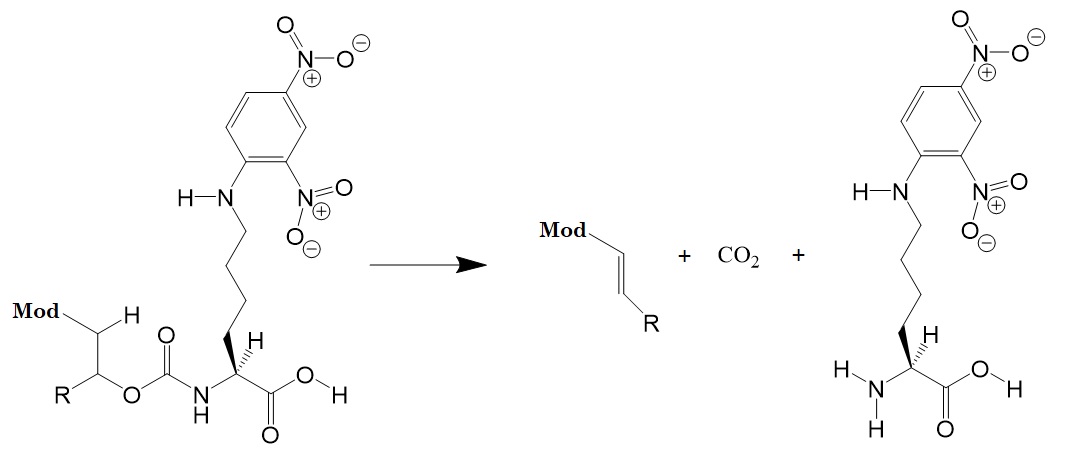
Figure 2: Releasing reaction of a linker scaffold containing electron-withdrawing pKa modulators linked via carbamate bonds to Ne-2,4-dinitrophenyl-l-lysine [Lys(DNP)OH]. Modulators utilized aromatic groups, ketones, nitriles, and sulfones.
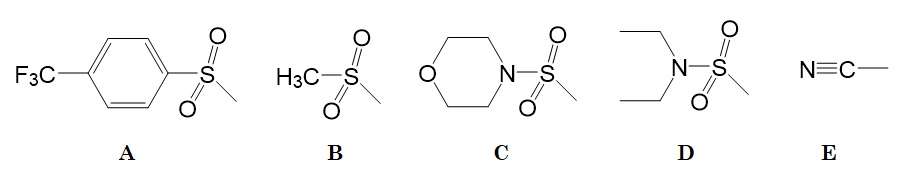
Figure 3: Modulator groups tested. [A] Half-lives for β-elimination varied between 14 hours to 2 weeks. [B to E] These less electron-withdrawing modulators extended the half-lives for up to 12 months (Santi et al. 2012).
Furthermore, the research group reported the preparation of bifunctional linkers containing succinimidyl carbonate on one end and an azide moiety connected to the other. After reacting the succinimidyl carbonate with the amino group of a carrier or a similar molecule, copper-free click chemistry allows conjugation to other desired functional groups. The reported heterobifunctional linkers allow attachment to the amine group of a drug or prodrug as a carbamate on one end and any macromolecular carrier on the other end.
The acidity of the ionizable C-H group is controlled by electron-withdrawing pKa modulators. After proton abstraction, the linkers undergo β-elimination to release the native, amine-containing drug or prodrug from the carrier molecules. Controlling the acidity of the ionizable C-H group by the pKa modulator allows for controlling the drug release rate.
Reference
Santi DV, Schneider EL, Reid R, Robinson L, Ashley GW. Predictable and tunable half-life extension of therapeutic agents by controlled chemical release from macromolecular conjugates. Proc Natl Acad Sci U S A. 2012 Apr 17;109(16):6211-6. [PMC]
Links
https://www.biosyn.com/oligo-modified-linker-attachment-chemsitry.aspx#!
https://www.biosyn.com/tew/oligonucleotide-linkers-for-conjugation.aspx#!
https://www.biosyn.com/oligonucleotideproduct/photocleavable-pc-linker.aspx
https://www.biosyn.com/tew/affinity-controlled-release-of-5-fluorouracil-by-synthetic-peptide-d-leu-phe-phe-derived-hydrogels-for-the-potential-treatment-of-skin-cancer.aspx
Pharmacokinetics NIH Library
---...---
" Bio-Synthesis provides custom synthesized GalNAc-conjugated oligonucleotides including GalNAc-siRNAs.
Bio-Synthesis also provides a full spectrum of high quality custom oligonucleotide modification services including back-bone modifications, conjugation to fatty acids, biotinylation by direct solid-phase chemical synthesis or enzyme-assisted approaches to obtain artificially modified oligonucleotides, such as BNA antisense oligonucleotides, mRNAs or siRNAs, containing a natural or modified backbone, as well as base, sugar and internucleotide linkages.
Bio-Synthesis also provides biotinylated and capped mRNA as well as long circular oligonucleotides".
---...---