Polyhistidine is a common and ubiquitous affinity tag used in expression vectors or protein expression and purification reagents. Many researchers use the polyhistidine tag with six histidines also known as the His-tag or His6 tag. However, the tag's length can vary between two (2) to ten (10) histidine residues per tag. Histidine readily forms coordination bonds with immobilized metal ions. The fusion of a tag to peptides or proteins allows efficient purification using affinity chromatography. Also, in-vivo tagged proteins enable functional imaging of protein locations in cells or tissue. Selecting sequences of gene fragments and clonal genes allow the design of desired fusion proteins using DNA sequences originating from natural or mutated proteins.
Since the purification of specific proteins from native sources is often very challenging, researchers take advantage of heterologous expression systems. In this approach, cloning genes into artificial vectors allow the expression of selected tagged proteins within easily cultured cells, such as Escherichia coli (bacteria), Pichia pastoris (yeast), and several insect and mammalian cells. The fusion of the protein of interest with a carrier protein or a peptide and protein tag can increase solubility, change the localization, allow affinity purification, or allow immunological assays.
His-Tags
|
TAG
|
INFO
|
SEQUENCE
|
|
|
|
|
|
His-tag
|
This mixed CAC/CAT oligonucleotide His-tag sequence is easier to synthesize than the low-diversity 6x CAT His-tag.
|
CAC CAT CAT CAC CAC CAT
H H H H H H
His-His-His-His-His-His
|
|
His peptide
|
This protein sequence was found to chelate divalent metals. The short recognition motif contains six histidines interspersed among other amino acids. Allows purification of semi-pure proteins in one step. The matrix used may be regenerated and reused almost infinitely. But this tag does not bind metal affinity resin as tightly as the His-tag. The tag or elution conditions can affect fusion properties.
|
KDHLIHNVHKEFHAHAHNK
KDHLIHNVHKEEHAHAHNK
(N-terminal end of MAP/microtubule affinity-regulating kinase 3)

 1v5s 1v5s
|
Immobilized metal affinity chromatography (IMAC) based on polyhistidine tracts' affinity for divalent cations allows purification of His-tag fusion proteins. Typically, resins with the immobilized nickel cation, Ni2+, chelate complex is utilized for affinity purification. The immobilized nickel ion, Ni2+, is most commonly used combined with the His-tag, but immobilized Co2+, Cu2+, Ni2+, Zn2+, Ca2+, and Fe3+ can all be used as well for the purification of polyhistidine fusion proteins.
IMAC allows the purification of recombinant proteins containing the tag as a fusion protein, either on the n-terminal or c-terminal end. The resin or matrix most widely used for IMAC is Ni(II)-nitrilotriacetic acid (Ni-NTA). However, other resins that also allow the immobilization of transition metal ions include iminodiacetic acid agarose and carboxy-methyl-aspartate agarose.
The chelating agent nitrilotriacetic acid (NTA) with the formula N(CH2CO2H)3 forms coordination complexes with metal ions, known as chelates, such as Ca2+, Co2+, Cu2+, Fe3+, Ni2+ and Zi2+.
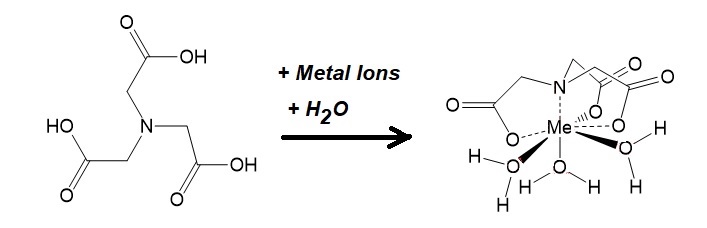
Figure 1: Chelation of metal ions by NTA.
Reference
B. L. Barnett, V. A. Uchtman "Structural investigations of calcium-binding molecules. 4. Calcium binding to aminocarboxylates. Crystal structures of Ca(CaEDTA).7H2O and Na(CaNTA)" Inorg. Chem., 1979, volume 18, pp 2674–2678. https://pubs.acs.org/doi/abs/10.1021/ic50200a007
Green and Sambrook; Molecular Cloning. A Laboratory Manual. 4th Edition. pp 1571-1577.
Kimple ME, Brill AL, Pasker RL. Overview of affinity tags for protein purification. Curr Protoc Protein Sci. 2013 Sep 24;73:9.9.1-9.9.23. doi: 10.1002/0471140864.ps0909s73. PMID: 24510596; PMCID: PMC4527311. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC4527311/
Kneusel R.E., Wulbeck M., Ribbe J. (2000) Detection and Immobilization of Proteins Containing the 6xHis Tag. In: Rapley R. (eds) The Nucleic Acid Protocols Handbook. Springer Protocols Handbooks. Humana Press, Totowa, NJ. https://doi.org/10.1385/1-59259-038-1:935
Terpe K. Overview of tag protein fusions: From molecular and biochemical fundamentals to commercial systems. Appl Microbiol Biotechnol. 2003;60:523–533.
---...---