In aging humans, the length of telomeres declines in dividing cells. Each time cells divide, telomeres can get shorter. When the telomeres are too short, the cells can no longer divide. The cells become inactive, or “senescent,” or die. The shortening process is associated with aging, cancer, and a high risk for death. Telomeres play a central role in cell fate and aging. Telomere repeats cap most chromosomes if not all to avoid activation of DNA repair pathways. Short telomeres are implicated in a variety of disorders. Telomeres shorten with physiological aging. However, during cancer immortalization telomeres undergo significant restoration. Determination of telomere lengths suggests that an age-based reference can be established for telomere studies. Therefore the availability and development of accurate and sensitive techniques and methods allowing measuring the lengths of telomeres in cells or cell tissue are needed.
Researchers at the UTSW Medical Center, Dallas, Texas have recently developed a method to measure telomere length.
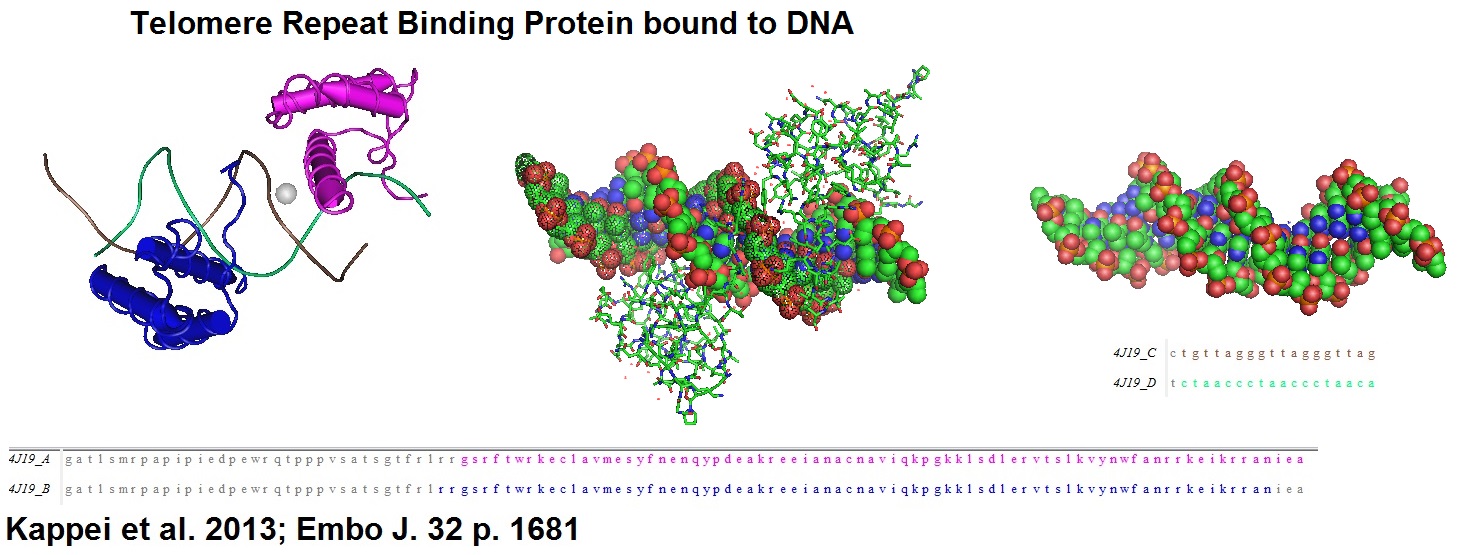
Figure 1: Structural models of homeobox telomere-binding protein 1 (HOT 1), a mammalian direct telomere repeat-binding protein. HOT1 is a positive regulator of telomere length that supports telomerase-depending telomere elongation.
A universal priming probe was used for TRFanalysis
5’-(Phos)GACTCTCAACTATC+T+A-3’
+N represents the location of BNAs.
Universal BNA Priming Probe
-.-
BNA Probes and Oligonucleotides can be ordered from Bio-Synthesis Inc.
-.-
BNA-digoxigenin-probes for enhanced telomere length analysis
BNA-oligonucleotide probes were designed to specifically bind to telomere repeats. For this, the researchers designed a non-radioactive labeling method that uses 3’ fill-in combined with lambda exonuclease digestion for the incorporation of one or more digoxigenin molecules into bridged nucleic acid (BNA)-containing oligonucleotides. Using this method, the researchers generated probes for the detection of both C- and G-rich telomeric DNA strands. The use of this type of probes enhanced the sensitivity of telomere length measurements significantly.
In humans, telomere lengths have been associated with cancer and age-related diseases. Telomeres are located at the ends of chromosomes and are composed of tandem 5’-TTAGGG-3’ repeats. Shelterin proteins associate with telomeres and play essential roles in telomere protection, telomerase regulation and the prevention of chromosome degradation. As humans age, telomeres gradually shorten in all dividing cells. The shortening triggers DNA damage responses and cellular senescence that can lead to genomic instability and cancer progression, especially if oncogenic changes in cells occur.
Telomere-specific probes used for Southern blotting in combination with a technique called “Terminal Restriction Fragment Analysis” enables the direct detection of different sizes of telomeres.
Terminal restriction fragment (TRF) pattern analysis, also known as “Terminal Restriction Fragment Length Polymorphisms (T-RFLP) analysis, is a recently developed PCR-based method. This technique also allows studying microbial community structure and dynamics.
How does the technique work?
1. T-RFLP analysis measures the size polymorphism of terminal restriction
fragments from PCR amplified markers.
2. Primers needed for this technique are designed with the help of comparative
genomics.
3. Primers are designed against the amplification product or amplicon.
4. PCR amplifies the signal from a high background of unreleated markers.
5. Subsequent digestion with correctly selected restriction endonucleases
produce terminal fragments.
6. Fragments are separated on high resolution sequencing gels.
7. A digital output is generated if separation is done in a capillary electrophoresis
system.
8. The use of fluorescently tagged or labeled primers limits the analysis to only
the terminal fragments of the digestion.
9. Using internal size markers with a different fluorophore makes the sizing
very accurate.
Outline of TRF analysis protocol
|
|
According to Lai et al. 2016.
|
|
Step 1
|
a. Prepare template DNA for DIG-labeled telomere C-rich (TC) or
G-rich (TG) probe synthesis.
b. Anneal G-rich or C-rich template oligonucleotides to a universal
priming oligonucleotide.
|
|
|
|
|
|
Templates
|
|
|
The 5’-phosphorylated template oligonucleotide begins with seven telomeric repeats followed by a short non-telomeric sequence.
|
|
G-rich template
|
5’-(Phos)CCC TAA CCC TAA CCC TAA CCC TAA CCC TAA CCC TAA CCC TAF ATA GTT GAG AGT C-3’
|
|
C-rich template
|
5’-(Phos)GGG TTA GGG TTA GGG TTA GGG TTA GGG TTA GGG TTA GGG TTA GAT AGT TGA GAG TC-3’
|
|
|
|
|
|
Universal priming
|
|
|
The universal priming oligonucleotide is phosphorylated at the 5’-end and contains a sequence complementary to the non-telomeric sequence in the template oligonucleotide with additional thymine (T) and adenine (A) at the 3’-end to ensure that it anneals at the correct spot.
Oligonucleotides modified with BNAs are used to increase resistance to nuclease digestion and the affinity for the target DNA or RNA.
|
|
Universal primer
|
5’(Phos)GAC TCT CAA CTA TC+T +A-3’; +N = BNAs
|
|
|
Use Exo- Klenow Fragment together with a dNTP mix containing DIG-11-dUTP (Roche Applied Sciences, Mannheim, Germany) for 3’ fill-in reactions.
|
|
|
Remove additional nucleotides from 3’-end
|
|
Step 2
|
Apply T4 DNA polymerase to remove additional nucleotides at the 3’ end from the template DNA generated by 3’ fill-in reactions. This increases the specificity of DIG-labeled telomere probes.
|
|
Step 3
|
Use λ exonuclease to digest the 5’-phosphorylated template oligonucleotide and non-telomeric sequence in the priming oligonucleotide (5’->3’ direction). Note: λ exonuclease is unable to degrade BNA-containing telomeric-specific single-stranded DNA.
|
|
Step 4 A
|
At this step a dot blot on a nylon membrane can be performed to check that the experiment worked. [See DIG Application Manual for filter hybridization.
|
|
Step 4B
|
Perform Southern blot analysis.
|
|
|
Southern Blot Analysis
|
|
Step 1
|
Digest DNA and DIG-labled molecular weight marker II.
|
|
Step 2
|
Separate on a 7% agarose gel.
|
|
Step 3
|
Depurinate, denature and neutralize the gel.
|
|
Step 4
|
Transfer DNA fragments onto a positive charged nylon membrane using a vacuum blotting system.
|
|
Step 5
|
Fix the DNA fragments on the membrane by UV-crosslinking.
|
|
Step 6
|
Pre-hybridize.
|
|
Step 7
|
Hybridize with DIG Easy Hyb solution containing one of the DIG-labeled telomeric probes over night.
|
|
Step 8
|
Wash membrane.
|
|
Step 9
|
Detect chemiluminescence signals.
|
|
Step 10
|
Analyze.
|
See Supplement “Material and methods” from Lai et al. for more details.
Example of DIG-probe synthesis
Start with templates and universal BNA probe(s):
Tandem 5’-TTAGGG-3’ repeat
GGGTTAGGGTTAGGGTTAGGGTTA...
5’(Phos)GACTCTCAACTATCTA-3’
3’-CTGAGAGTTGATAGATCCCAATCCCAATCCCAATCCCAAT...N-PHOS
->Anneal -> 3’fill-in (Exo- KF) -> DNA blunting (T4 DNA polymerase) -> 5 ->3’ digestion (λ exonuclease) -> DIG probes.
The use of fluorescently labeled oligonucleotide probes instead of 32P-labeled oligonucleotides makes this technique more convenient and less hazardous since many waste disposal and safety issues are associated with radioactivity.
Reference
DIG RNA Labeling Kit (SP6/T7): http://www.sigmaaldrich.com/catalog/product/roche/11175025910?lang=en®ion=US
Kyung H. Choi, Amy S. Farrell, Amanda S. Lakamp, Michel M. Ouellette; Characterization of the DNA binding specificity of Shelterin complexes. Nucleic Acids Res. 2011 Nov; 39(21): 9206–9223. Published online 2011 Aug 18. doi: 10.1093/nar/gkr665, PMCID: PMC3241663.
Raffaella Diotti, Diego Loayza; Shelterin complex and associated factors at human telomeres. Nucleus. 2011 Mar-Apr; 2(2): 119–135. doi: 10.4161/nucl.2.2.15135, PMCID: PMC3127094.
Kong PL, Looi LM, Lau TP, Cheah PL.; Assessment of Telomere Length in Archived Formalin-Fixed, Paraffinized Human Tissue Is Confounded by Chronological Age and Storage Duration. PLoS One. 2016 Sep 6;11(9):e0161720. doi: 10.1371/journal.pone.0161720. eCollection 2016.
Aubert G, Lansdorp PM.; Telomeres and aging. Physiol Rev. 2008 Apr;88(2):557-79. doi: 10.1152/physrev.00026.2007.
Lai, Tsung-Po, Wright, Woodring E., and Shay, Jerry W. ; 2016. Generation of digoxigenin-incorporated probes to enhance DNA detection sensitivity. BioTechniques 60:306-309 (June 2016).
Marsh TL; Terminal restriction fragment length polymorphism (T-RFLP): an emerging method for characterizing diversity among homologous populations of amplification products. Curr Opin Microbiol. 1999 Jun;2(3):323-7.
Dennis Kappei, Falk Butter, Christian Benda, Marion Scheibe, Irena Draškovi?, Michelle Stevense, Clara Lopes Novo, Claire Basquin, Masatake Araki, Kimi Araki, Dragomir Blazhev Krastev, Ralf Kittler, Rolf Jessberger, J Arturo Londoño?Vallejo, Matthias Mann, Frank Buchholz; HOT1 is a mammalian direct telomere repeat?binding protein contributing to telomerase recruitment. The EMBO Journal (2013) 32, 1681-1701.
-.-